Respiratory Viral Coinfections: Impacts on Virus Replication and Host Immune Response
Abstract
Keywords:
Respiratory, viral, infectionHighlights
Coinfection outcomes are driven by the sequence of infection and innate immune activation.
Type I/III interferons establish antiviral states that can suppress secondary viral replication under certain conditions.
IL-1-mediated inflammation amplifies lung injury and may represent a therapeutic target.
Respiratory viruses can compete, cooperate, or hybridize, shaping viral evolution and severity.
Mechanistic mapping clarifies contradictory clinical observations across coinfections.
Introduction
Coinfection is common among respiratory viral infections and can result in severe symptoms in affected patients (19). The global prevalence of viral coinfection is still unknown; however, interest in this topic has increased in recent years. Studies have estimated a coinfection rate of 5.01% among patients with coronavirus disease 2019 (COVID-19), with higher prevalence reported in children (9.39%) (2). One cohort study reported a coinfection rate of 97.2% in pediatric cases (3). Although coinfection has a wide range of prevalence, most studies indicate that coinfection rates are higher in children.
There are several types of viral coinfections, including homologous coinfection, which refers to the simultaneous infection of two different viruses from the same viral family; heterotypic coinfection, which results from the simultaneous infection of two viruses within the same viral species; and heterologous coinfection, which results from the simultaneous infection of two different viruses from different families (4). Respiratory virus co-circulation is one of the important reasons for coinfection. Co-circulation refers to the concurrent circulation and transmission of multiple respiratory viruses within the human population (4). Coinfection is usually observed during winter and cold months and is defined as infection with two or more viruses simultaneously or a secondary viral infection occurring shortly after a previous infection (4). Viral coinfection can influence viral pathogenicity, immune responses, and disease symptom (5).
In particular, coinfection with respiratory viruses such as influenza and COVID-19 can lead to more severe symptoms, complicate treatment, and place a greater burden on healthcare staff (6). Coinfection is particularly important in respiratory viral infections because these viruses can easily cause epidemics or pandemics, as demonstrated by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic. Coinfections may lead to more extensive damage to the respiratory tract epithelium and lung alveoli. This damage leads to increased release of damage-associated molecular patterns (DAMPs), such as adenosine triphosphate (ATP) and mitochondrial DNA. These molecules themselves are powerful stimulators of inflammasome activation and interleukin-1 (IL-1) production (7). Inflammation is mediated by pro-inflammatory cytokines, including IL-1, IL-6, tumor necrosis factor (TNF), and IL-8. IL-1 is the most studied cytokine with properties relevant to several inflammatory diseases, including viral infections (8). Viral coinfection usually alters immune responses and sometimes influences the course of the second infection. As summarized in Table 1, interferon-mediated viral interference and order-dependent immune modulation are emerging as key mechanisms governing respiratory viral coinfections (9). Some studies have shown that coinfection in COVID-19 patients is associated with increased disease severity and mortality, decreased lymphocyte counts, and reduced numbers of cluster of differentiation 4-positive (CD4+) T cells, CD8+ T cells, and B cells (10). In this review, we synthesize current evidence regarding respiratory viral coinfections and examine their effects on viral replication and immune responses.
Influenza and SARS-CoV-2 Coinfection
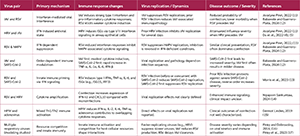
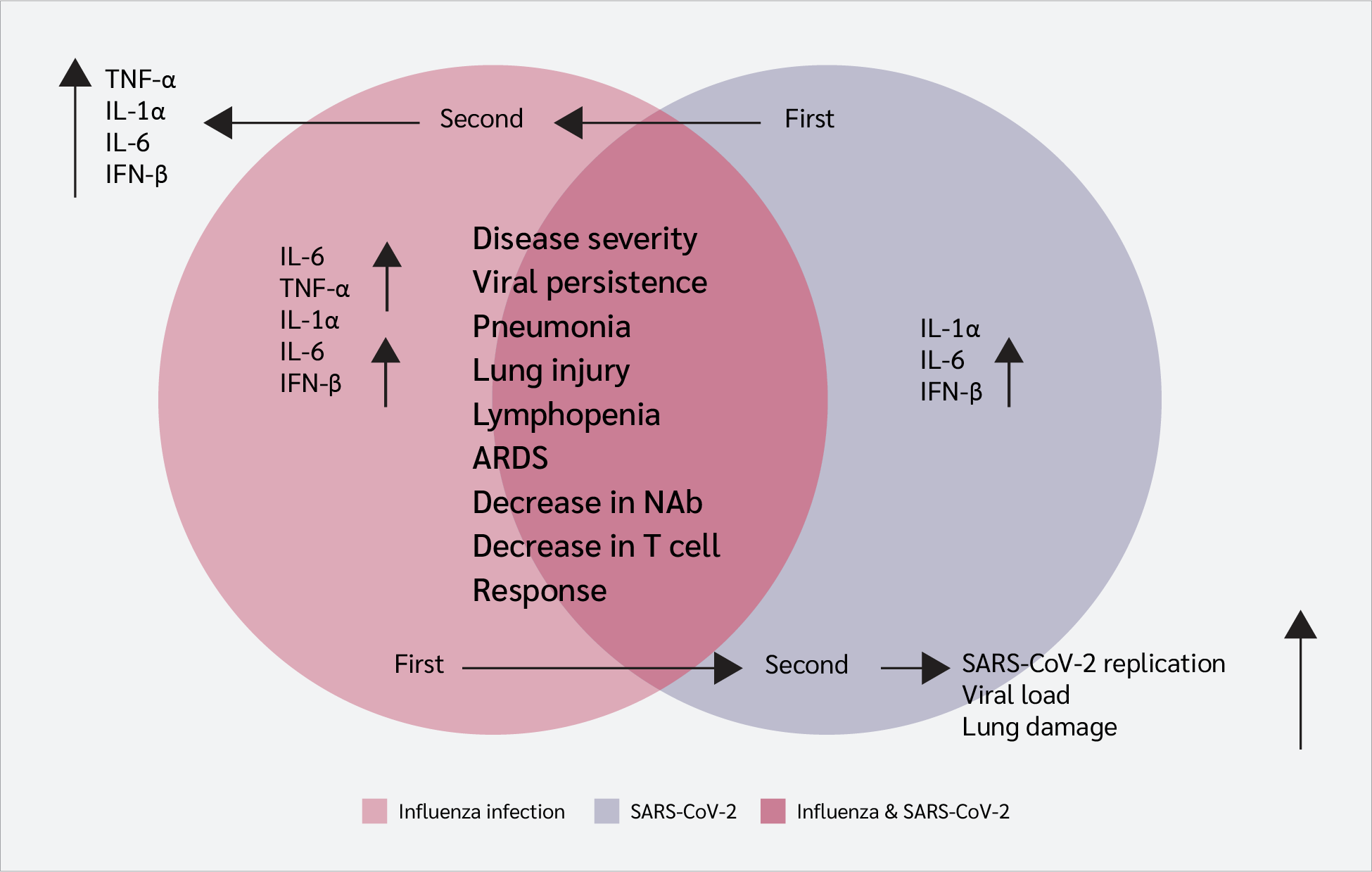
Studies have shown that the influenza A virus is more frequently associated with SARS-CoV-2 coinfection than influenza virus type B (6). In hamsters and mice, influenza virus infection increases levels of IL-6, TNF-α, IL-1α, and interferon-β (IFN-β). Infection with SARS-CoV-2 leads to increased levels of IL-1α, IL-6, and IFN-β, although these levels are lower than those observed in a single infection with influenza A virus. Influenza infection after SARS-CoV-2 causes increased levels of TNF-α, IL-1α, IL-6, and IFN-β (Figure 1) (12).
Studies investigating influenza and SARS-CoV-2 coinfection in hACE2 transgenic mice have shown increased disease severity, viral persistence in the lungs, pneumonia, and lung injury. One study evaluated innate immune responses in influenza and SARS-CoV-2 coinfection in human respiratory tissue explants and human airway and alveolar epithelial cells. Coinfections resulted in a significant upregulation of innate immune responses to SARS-CoV-2 compared with single infections and led to more severe host damage (18).
Coinfection of influenza and SARS-CoV-2 has been shown to induce lymphopenia, which ultimately impairs adaptive immune responses and reduces levels of neutralizing antibodies and T-cell responses (19). Influenza virus infection before SARS-CoV-2 has been reported to suppress SARS-CoV-2 replication (20).
Coinfection with these viruses stimulates activation of inflammatory immune responses, increases immunopathology, and contributes to lung damage and acute respiratory distress syndrome (ARDS) (21). However, another study found that pre-infection with influenza enhanced SARS-CoV-2 replication, viral load, and lung damage (22).
Immune responses to the first infection (influenza virus in this case) can affect inflammatory responses to SARS-CoV-2 and play an auxiliary role in viral infectivity. It is possible that immune responses to the first infection facilitate receptor access for the second virus. On the other hand, a viral infection has a confounding effect on another one. The results obtained in various studies are inconsistent; however, sequential influenza and SARS-CoV-2 infections influence each other's progression.
Influenza and Respiratory Syncytial Virus Coinfection
In mice, respiratory syncytial virus (RSV) infection has been shown to lead to protection against subsequent influenza infection (12). Influenza infection prior to RSV caused airway dysfunction and weight loss, whereas RSV infection prior to influenza was associated with lower mortality and morbidity. Levels of TNF, IL-6, and interferon-γ (IFN-γ) were elevated in coinfection groups compared with RSV control groups. Tumor necrosis factor and IL-6 levels were decreased in coinfection groups compared with influenza control groups. This study also showed that the protective effect of RSV caused a decrease in influenza immunopathology, which may be due to increased levels of IFN-γ after RSV infection (23). Another study showed the protective effect of influenza against RSV infection lasting up to 1–5 months after recovery (24).
Additionally, a study confirmed that influenza suppresses RSV replication by achieving a higher growth rate compared with RSV (16). In mice, RSV infection can protect against influenza infection and reduce inflammatory responses during influenza virus infection (12).
In this context, competitive conditions inhibit RSV replication. The possibility of using this feature to inhibit the replication of dangerous viruses by leveraging high-replication, low-risk viruses should be investigated.
A study found that RSV and influenza coinfection can lead to the formation of hybrid viruses. These hybrid viral particles can express several proteins from both parental viruses (25). The ability to integrate and produce new viruses during coinfection may lead to the emergence of dangerous viruses with the potential for global pandemics. Given the potential to produce new infectious viral particles with high replication capacity and pathogenicity, especially in respiratory viruses, this hypothesis should be carefully examined.
Influenza Coinfection with Adenovirus and Rhinovirus
Influenza and adenovirus coinfection has been shown to increase levels of inflammatory cytokines such as IL-6 and IL-1α in the A549 lung cell line (26).
Studies have shown that rhinovirus infection reduces influenza virus replication during subsequent infection. Interferon responses stimulated by rhinovirus infection in airway epithelial cells play a protective role against influenza infection (27). Another study revealed the protective effect of rhinovirus against subsequent influenza infection in human bronchial epithelial cells (HBECs) and human nasal epithelial cells (HNECs) (28).
Influenza A (H1N1) infection in airway cells was found to suppress rhinovirus replication, whereas rhinovirus did not change H1N1 replication in airway epithelial cells (29).
Influenza and Parainfluenza Coinfection
A study showed that primary infection with parainfluenza viruses enhances influenza virus replication in respiratory epithelial cells (30). Human parainfluenza virus type 2 has a booster effect on cell fusion in epithelial cells. Parainfluenza virus induced cell fusion and increased access to sialic acid for influenza hemagglutinin (HA) antigen, thereby increasing influenza virus infectivity (30).
SARS-CoV-2 and RSV Coinfection
Respiratory syncytial virus and SARS-CoV-2 coinfection, particularly when RSV infection precedes SARS-CoV-2, has been associated with decreased disease severity and reduced SARS-CoV-2 replication in BALB/c mice models (13). SARS-CoV-2 coinfection has also been shown to enhance RSV replication.
Conversely, when RSV infection occurs after SARS-CoV-2 infection, reduced RSV replication and protection have been observed (13). Previous RSV infection appeared to have a protective effect against subsequent SARS-CoV-2 infection in BALB/c mice (31). This effect may be due to persistent interferon responses after RSV infection.
SARS-CoV-2 and Adenovirus Coinfection
In vitro studies on SARS-CoV-2 and adenovirus have shown that these viruses do not affect each other’s replication in Vero E6 cells. However, hamsters coinfected with these viruses exhibited more severe lung damage and clinical symptoms.
Inflammatory immune responses, including interferon, IL-6, CC motif chemokine ligand 17 (CCL17), and transforming growth factor-β (TGF-β), remained elevated for a prolonged period after infection in coinfected animals compared with those with single infections. Notably, IL-6 levels were markedly increased in coinfected animals, which may explain the severe symptoms and lung damage in this condition (32).
In another study, levels of IL-6, IL-1, and interferon-α (IFN-α) were significantly elevated in A549 cells coinfected with SARS-CoV-2 and adenovirus compared with adenovirus mono-infection 3 days after infection (26).
SARS-CoV-2 and Rhinovirus Coinfection
A study indicated that SARS-CoV-2 replication decreases after rhinovirus infection. However, when SARS-CoV-2 infection occurs first, rhinovirus replication does not change (28).
SARS-CoV-2, RSV, and Pneumoviridae Virus Coinfection
Studies have shown that prior human metapneumovirus (hMPV) infection increases lung susceptibility to SARS-CoV-2 infection (33). Respiratory syncytial virus infection has been shown to inhibit hMPV replication, with interferon responses playing a central role.
It is difficult to distinguish between RSV and hMPV infection or coinfection because both viruses present similarly, including severe bronchiolitis. Studies have shown that hMPV viral load is higher in mono-infection than in coinfection with RSV, although this pattern is not observed in RSV mono-infection. Interferon responses against RSV may reduce hMPV replication (12,34).
Immune Responses
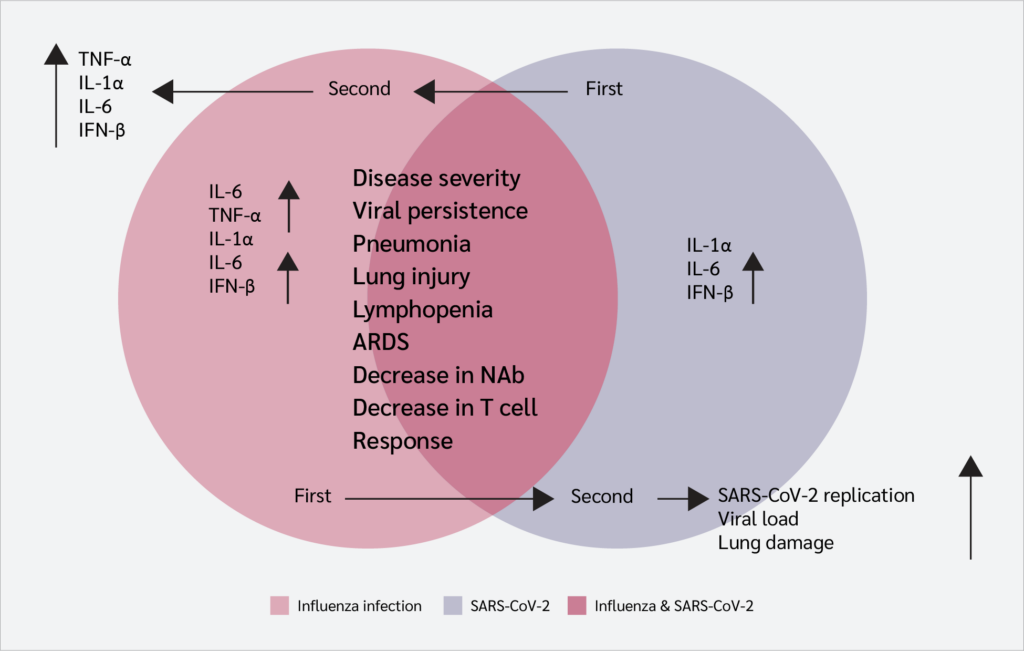
Immune responses triggered by an initial infection can shape the host’s response to a subsequent infectious challenge. In respiratory infections, especially those caused by respiratory viruses, interferons are among the earliest immune signals produced and place host cells into an antiviral state while inducing the expression of pro-inflammatory genes (35,36).
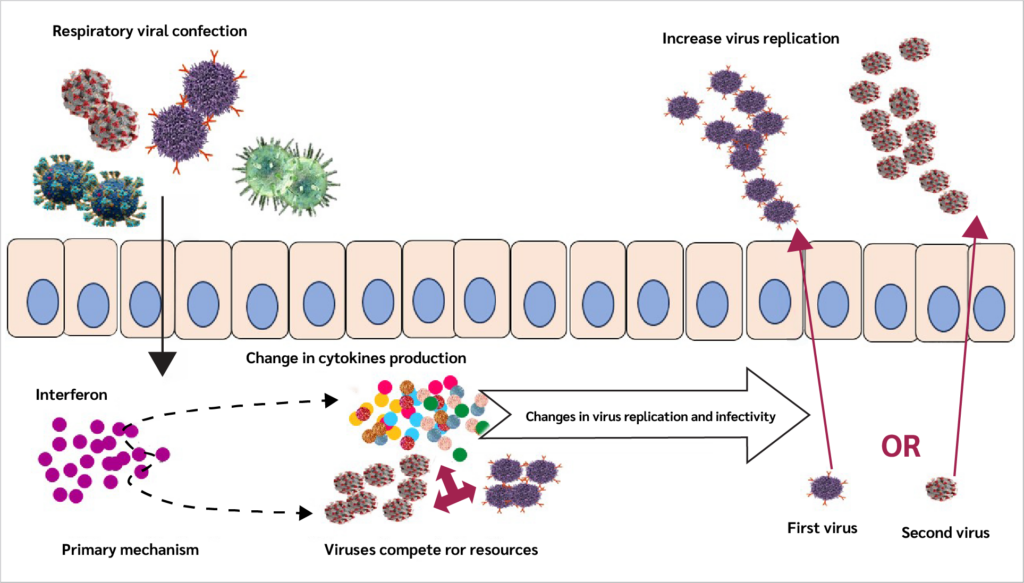
These changes in gene expression influence multiple aspects of the local environment, including mucus production, extracellular and intracellular conditions, and immune cell activity. As a result, understanding the impact of a primary infection on immunity to a secondary infection requires consideration of several interconnected factors, such as alterations in viral receptor expression, viral entry and replication, viral gene expression, competition for host cellular resources, enhancement of inflammatory responses, and mechanisms that regulate or limit immune activation (Figure 2).
Additionally, IL-1 production is important because it is considered a key mediator of immunity and inflammation (37,38). For example, during influenza virus infection, IL-1 induces trypsin upregulation (39), and trypsin is necessary for viral fusion during influenza virus entry (40). These findings suggest that IL-1 may facilitate influenza infectivity; accordingly, viral infections that elevate IL-1 levels could enhance influenza virus entry and fusion.
Interleukin-1 enhances the recruitment of neutrophils and macrophages to the lungs, and these cells increase tissue damage by releasing enzymes and free radicals, potentially leading to more severe ARDS (41). Interleukin-1 also increases vascular permeability, leading to edema (fluid accumulation) in lung tissue, which further impairs gas exchange. (35)
Therefore, interleukin-1β (IL-1β) is an attractive potential therapeutic target for intervention in severe cases of these coinfections (42). Nevertheless, we have only briefly mentioned the impact of immune responses, such as IL-1, in this context. The effects of these cytokines in coinfections with different viruses should be further studied, and such studies may help identify new therapeutic targets.
Immune responses during respiratory viral infections represent a double-edged sword. While they help clear the virus, they sometimes go too far and end up damaging tissue instead. For instance, overproduction of inflammatory cytokines like IFN-γ has been linked to chronic obstructive pulmonary disease (COPD), ARDS, and even fatal pneumonia (43).
In one study, ferrets were first infected with RSV and then with influenza A virus (IAV) 3 days later. The findings demonstrated that IAV slowed down RSV replication, whereas RSV seemed to delay the clearance of IAV-infected cells (17).
When multiple viruses infect the same host, their interactions can vary—they may enhance one another, interfere with each other, or have no noticeable effect at all. These interactions vary depending on which viruses are involved, highlighting the importance of understanding what is happening at the molecular level (12). A deeper characterization of virus-virus and virus-host interactions is therefore essential not only for predicting disease outcomes but also for developing better treatments that reduce tissue damage while still clearing the infection.
Conclusion
Respiratory viral coinfection represents a significant challenge in respiratory infectious diseases. Such coinfections can affect immune responses and result in the emergence of new viruses. The immune response and disease severity may vary when the immune system faces several viral infections simultaneously.
This condition may facilitate the generation of new viral strains that are sometimes more infectious. Coinfection can also affect viral replication. However, differences in experimental models, infection sequence, viral dose, and host species likely explain the contradictory outcomes observed across studies.
For some viruses, prior infection or coinfection can act like a vaccine, reducing viral replication. Many hypotheses have been proposed regarding coinfection, including its role in the emergence of new viruses, changes in viral infectivity, and disease symptoms. The hypothesis of using one virus as a vaccine against another, through modulation of immune responses, also warrants further investigation.
Considering these aspects, coinfection is particularly relevant during viral pandemics and in immunocompromised patients. Therefore, investigating the impact of viral coinfections can be considered a goal to prevent further mortality and to help restrain viral infections.
Future Directions
Future research should focus on the molecular pathways of virus-virus and virus-host interactions and understanding these mechanisms at the single-cell level in human tissues.
Large-scale, longitudinal cohort studies are needed to translate experimental findings into clinically relevant insights for human populations.
Investigations should explore whether attenuated viral vectors or engineered stimuli of specific innate immune pathways (e.g., controlled induction of IFN-λ) can be developed as prophylactic or early intervention strategies to mitigate subsequent severe infections.
Surveillance programs and experimental studies should assess the risk of coinfection as a catalyst for the emergence of viruses with altered transmissibility, pathogenicity, or antigenic profiles.
Ethical Approval
Not applicable
Informed Consent
Not applicable
Peer-review
Externally peer-reviewed
Author Contributions
Concept – Z.K., F.S., A.G., S.F.; Design – A.M., S.I.D., A.K.; Supervision – A.K., S.F., Z.K.; Literature Review – A.N., J.F., A.M., F.S., S.I.D.; Writer – Z.K., A.G., F.S., A.M.; Critical Reviews – A.K., S.F., S.I.D., A.N., J.F.; Other – A.G., A.N., J.F.
Conflict of Interest
The authors declare no conflict of interest.
Financial Disclosure
The authors declared that this study has no financial support.
Acknowledgment
The authors thank the anonymous reviewers for their insightful comments and constructive suggestions, which improved the quality of the manuscript. The authors also thank the editorial team of the Turkish Journal of Immunology for their professional support and assistance throughout the publication process.
AI Statement
Not applicable
References
Georgakopoulou VE. Insights from respiratory virus co-infections. World J Virol. 2024;13(4):98600. [CrossRef]
Krumbein H, Kümmel LS, Fragkou PC, Thölken C, Hünerbein BL, Reiter R, et al. Respiratory viral co-infections in patients with COVID-19 and associated outcomes: A systematic review and meta-analysis. Rev Med Virol. 2023;33(1):e2365. [CrossRef]
Fu C, Huang Q, Zhao J, Mo L, Tang W, Lu J, et al. Clinical characteristics and co-infection analysis of influenza a virus in pediatric respiratory infections: a study based on tNGS technology. Eur J Clin Microbiol Infect Dis. 2025;44(7):1695–704. [CrossRef]
Trepat K, Gibeaud A, Trouillet-Assant S, Terrier O. Exploring viral respiratory coinfections: Shedding light on pathogen interactions. PLoS Pathog. 2024;20(9):e1012556. [CrossRef]
Du Y, Wang C, Zhang Y. Viral coinfections. Viruses. 2022;14(12):2645.
Golpour M, Jalali H, Alizadeh-Navaei R, Talarposhti MR, Mousavi T, Ghara AAN. Co-infection of SARS-CoV-2 and influenza A/B among patients with COVID-19: a systematic review and meta-analysis. BMC Infect Dis. 2025;25(1):145. [CrossRef]
Alpkvist H. Damage-associated molecular patterns and pathogen-associated molecular patterns in severe bacterial infections [doctoral thesis]. Stockholm (SE): Karolinska Institutet; 2024.
Conti P, Ronconi G, Caraffa A, Gallenga CE, Ross R, Frydas I, et al. Induction of pro-inflammatory cytokines (IL-1 and IL-6) and lung inflammation by Coronavirus-19 (COVID-19 or SARS-CoV-2): anti-inflammatory strategies. J Biol Regul Homeost Agents. 2020;34(2):327–31. [CrossRef]
Kumar N, Sharma S, Barua S, Tripathi BN, Rouse BT. Virological and immunological outcomes of coinfections. Clin Microbiol Rev. 2018;31(4):e00111–17. [CrossRef]
Zhang G, Zhang J, Gao Q, Zhao Y, Lai Y. Clinical and immunologic features of co-infection in COVID-19 patients, along with potential traditional Chinese medicine treatments. Front Immunol. 2024;15:1357638. [CrossRef]
Piret J, Boivin G. Viral interference between respiratory viruses. Emerg Infect Dis. 2022;28(2):273–81. [CrossRef]
Babawale PI, Guerrero-Plata A. Respiratory viral coinfections: insights into epidemiology, immune response, pathology, and clinical outcomes. Pathogens. 2024;13(4):316. [CrossRef]
Morris DR, Qu Y, Thomason KS, de Mello AH, Preble R, Menachery VD, et al. The impact of RSV/SARS-CoV-2 co-infection on clinical disease and viral replication: insights from a BALB/c mouse model. bioRxiv [Preprint]. May 24, 2023:2023.05.24.542043. [CrossRef]
Sankuntaw N, Punyadee N, Chantratita W, Lulitanond V. Coinfection with respiratory syncytial virus and rhinovirus increases IFN-λ1 and CXCL10 expression in human primary bronchial epithelial cells. New Microbiol. 2024;47(1):60–7.
Linden D, Guo-Parke H, Coyle PV, Fairley D, McAuley DF, Taggart CC, et al. Respiratory viral infection: a potential "missing link" in the pathogenesis of COPD. Eur Respir Rev. 2019;28(151):180063. [CrossRef]
Pinky L, Dobrovolny HM. Coinfections of the respiratory tract: viral competition for resources. PLoS One. 2016;11(5):e0155589. [CrossRef]
Pinky L, DeAguero JR, Remien CH, Smith AM. How interactions during viral-viral coinfection can shape infection kinetics. Viruses. 2023;15(6):1303. [CrossRef]
Ho JCW, Ng K, Ching RHH, Peiris M, Nicholls JM, Chan MCW, et al. Dynamic interaction between SARS-CoV-2 and influenza a virus infection in human respiratory tissues and cells. Microorganisms. 2025;13(5):988. [CrossRef]
Kim EH, Nguyen TQ, Casel MAB, Rollon R, Kim SM, Kim YI, et al. Coinfection with SARS-CoV-2 and influenza A virus increases disease severity and impairs neutralizing antibody and CD4+ T cell responses. J Virol. 2022;96(6):e0187321. [CrossRef]
Oishi K, Horiuchi S, Minkoff JM, tenOever BR. The host response to influenza A virus interferes with SARS-CoV-2 replication during coinfection. J Virol. 2022;96(15):e0076522. [CrossRef]
Swets MC, Russell CD, Harrison EM, Docherty AB, Lone N, Girvan M, et al. SARS-CoV-2 co-infection with influenza viruses, respiratory syncytial virus, or adenoviruses. Lancet. 2022;399(10334):1463–4. [CrossRef]
Bai L, Zhao Y, Dong J, Liang S, Guo M, Liu X, et al. Coinfection with influenza A virus enhances SARS-CoV-2 infectivity. Cell Res. 2021;31(4):395–403. [CrossRef]
Hartwig SM, Miller AM, Varga SM. Respiratory syncytial virus provides protection against a pubsequent influenza A virus infection. J Immunol. 2022;208(3):720–31. [CrossRef]
Kramer SC, Pirikahu S, Casalegno JS, Domenech de Cellès M. Characterizing the interactions between influenza and respiratory syncytial viruses and their implications for epidemic control. Nat Commun. 2024;15(1):10066. [CrossRef]
Haney J, Vijayakrishnan S, Streetley J, Dee K, Goldfarb DM, Clarke M, et al. Coinfection by influenza A virus and respiratory syncytial virus produces hybrid virus particles. Nat Microbiol. 2022;7(11):1879–90. [CrossRef]
Stincarelli MA, Arvia R, Guidotti B, Giannecchini S. Respiratory virus-specific and time-dependent interference of adenovirus type 2, SARS-CoV-2 and influenza virus H1N1pdm09 during viral dual co-infection and superinfection in vitro. Viruses. 2024;16(12):1947. [CrossRef]
Wu A, Mihaylova VT, Landry ML, Foxman EF. Interference between rhinovirus and influenza A virus: a clinical data analysis and experimental infection study. Lancet Microbe. 2020;1(6):e254–62. [CrossRef]
Essaidi-Laziosi M, Alvarez C, Puhach O, Sattonnet-Roche P, Torriani G, Tapparel C, et al. Sequential infections with rhinovirus and influenza modulate the replicative capacity of SARS-CoV-2 in the upper respiratory tract. Emerg Microbes Infect. 2022;11(1):412–23. [CrossRef]
Essaidi-Laziosi M, Geiser J, Huang S, Constant S, Kaiser L, Tapparel C. Interferon-dependent and respiratory virus-specific interference in dual infections of airway epithelia. Sci Rep. 2020;10(1):10246. Erratum in: Sci Rep. 2020;10(1):12523. [CrossRef]
Goto H, Ihira H, Morishita K, Tsuchiya M, Ohta K, Yumine N, et al. Enhanced growth of influenza A virus by coinfection with human parainfluenza virus type 2. Med Microbiol Immunol. 2016;205(3):209–18. [CrossRef]
Hartwig SM, Odle A, Wong L-YR, Meyerholz DK, Perlman S, Varga SM. Respiratory syncytial virus infection provides protection against severe acute respiratory syndrome coronavirus challenge. J Virol. 2024;98(9):e0066924. [CrossRef]
Svyatchenko VA, Ternovoi VA, Lutkovskiy RY, Protopopova EV, Gudymo AS, Danilchenko NV, et al. Human adenovirus and influenza a virus exacerbate SARS-CoV-2 infection in animal models. Microorganisms. 2023;11(1):180. [CrossRef]
Hashemi SA, Safamanesh S, Ghasemzadeh-Moghaddam H, Ghafouri M, Mohajerzadeh-Heydari MS, Namdar-Ahmadabad H, et al. Report of death in children with SARS-CoV-2 and human metapneumovirus (hMPV) coinfection: Is hMPV the trigger? J Med Virol. 2021;93(2):579–81. [CrossRef]
Talah S, Carbonneau J, Hamelin ME, Gilca R, Boivin G. Viral loads of pneumoviruses: correlation with coinfection rates and disease severity. J Med Virol. 2024;96(11):e70054. [CrossRef]
Fahey E, Doyle SL. IL-1 family cytokine regulation of vascular permeability and angiogenesis. Front Immunol. 2019;10:1426. [CrossRef]
Ivashkiv LB, Donlin LT. Regulation of type I interferon responses. Nat Rev Immunol. 2014;14(1):36–49. [CrossRef]
Makaremi S, Asgarzadeh A, Kianfar H, Mohammadnia A, Asghariazar V, Safarzadeh E. The role of IL-1 family of cytokines and receptors in pathogenesis of COVID-19. Inflamm Res. 2022;71(7-8):923–47. [CrossRef]
Mantovani A, Dinarello CA, Molgora M, Garlanda C. Interleukin-1 and related cytokines in the regulation of inflammation and immunity. Immunity. 2019;50(4):778–95. [CrossRef]
Indalao IL, Sawabuchi T, Takahashi E, Kido H. IL-1β is a key cytokine that induces trypsin upregulation in the influenza virus-cytokine-trypsin cycle. Arch Virol. 2017;162(1):201–11. Erratum in: Arch Virol. 2018;163(12):3487. [CrossRef]
Li ZN, Lee BJ, Langley WA, Bradley KC, Russell RJ, Steinhauer DA. Length requirements for membrane fusion of influenza virus hemagglutinin peptide linkers to transmembrane or fusion peptide domains. J Virol. 2008;82(13):6337–48. [CrossRef]
Dang W, Tao Y, Xu X, Zhao H, Zou L, Li Y. The role of lung macrophages in acute respiratory distress syndrome. Inflamm Res. 2022;71(12):1417–32. [CrossRef]
Hosseinian N, Cho Y, Lockey RF, Kolliputi N. The role of the NLRP3 inflammasome in pulmonary diseases. Ther Adv Respir Dis. 2015;9(4):188–97. [CrossRef]
Kombe Kombe AJ, Fotoohabadi L, Gerasimova Y, Nanduri R, Lama Tamang P, Kandala M, et al. The role of inflammation in the pathogenesis of viral respiratory infections. Microorganisms. 2024;12(12):2526. [CrossRef]
VOLUME
,
ISSUE
Correspondence
Received
Accepted
Published
Suggested Citation
DOI
License