A Preliminary Study on Soluble Immune Checkpoint Proteins in Systemic Lupus Erythematosus and Sjögren's Syndrome Patients
Abstract
Objective:
The aim of this study was to assess serum levels of soluble immune checkpoint proteins (sICPs) in patients with systemic lupus erythematosus (SLE) and Sjögren’s syndrome (SS) and to explore their association with disease activity.
Materials and Methods:
This preliminary cross-sectional study included 27 patients with SLE, 23 patients with SS, and 23 healthy controls. Serum concentrations of sCD25 (soluble interleukin-2 receptor alpha [IL-2Rα; CD25]), 4-1BB (tumor necrosis factor receptor superfamily member 9 [TNFRSF9; CD137]), B7.2 (CD86; cluster of differentiation 86), transforming growth factor beta 1 (TGF-β1), cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), programmed cell death protein 1 (PD-1), programmed death-ligand 1 (PD-L1), T-cell immunoglobulin and mucin-domain containing-3 (TIM-3), lymphocyte activation gene 3 (LAG-3), and Galectin-9 (Gal-9) were measured using a multiplex bead-based flow cytometric assay. Disease activity was evaluated using standard indices (Systemic Lupus Erythematosus Disease Activity Index [SLEDAI] for SLE; EULAR Sjögren’s Syndrome Disease Activity Index [ESSDAI] for SS). Group differences were analyzed, and correlations between sICP levels and clinical indices were assessed.
Results:
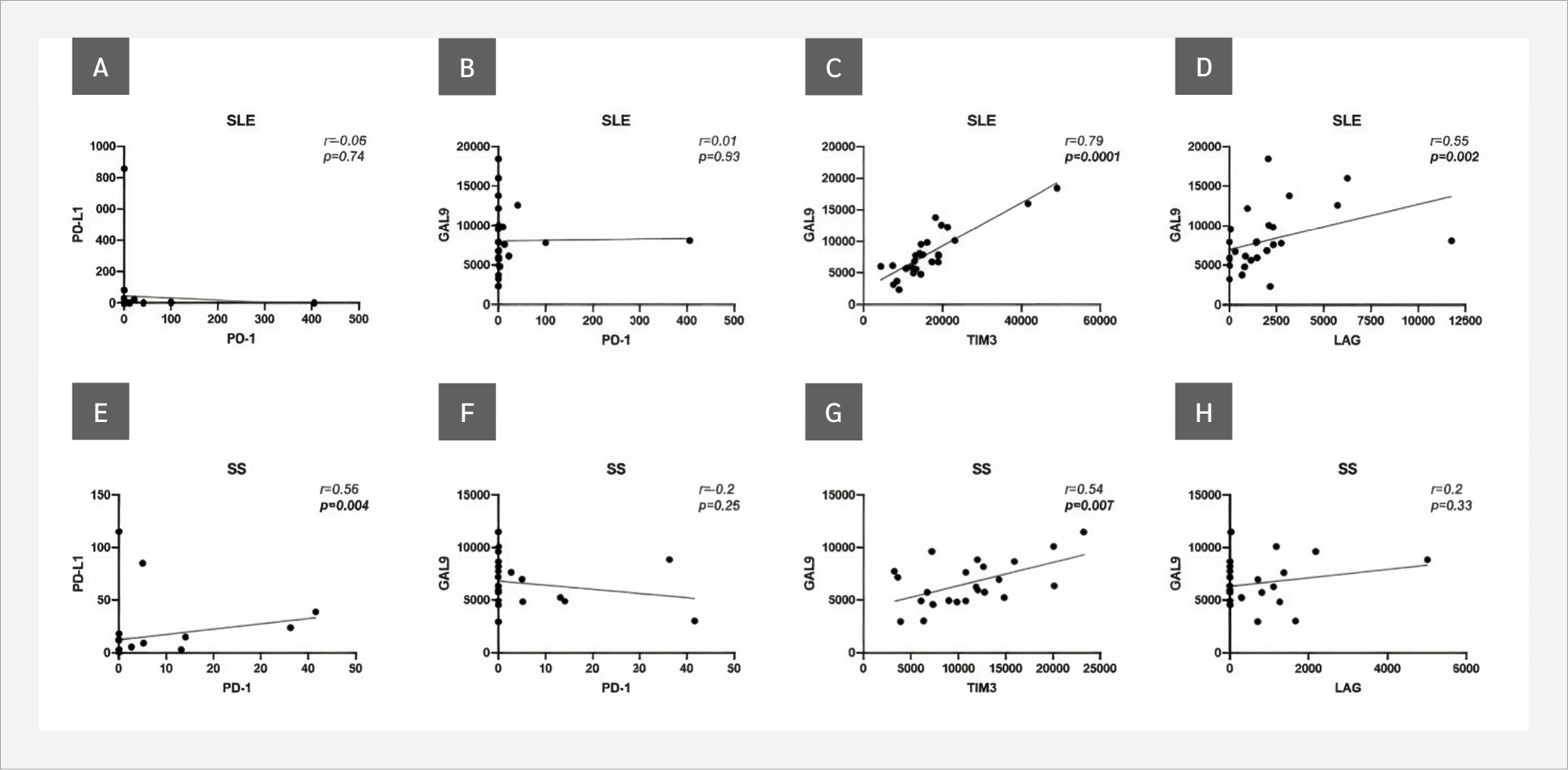
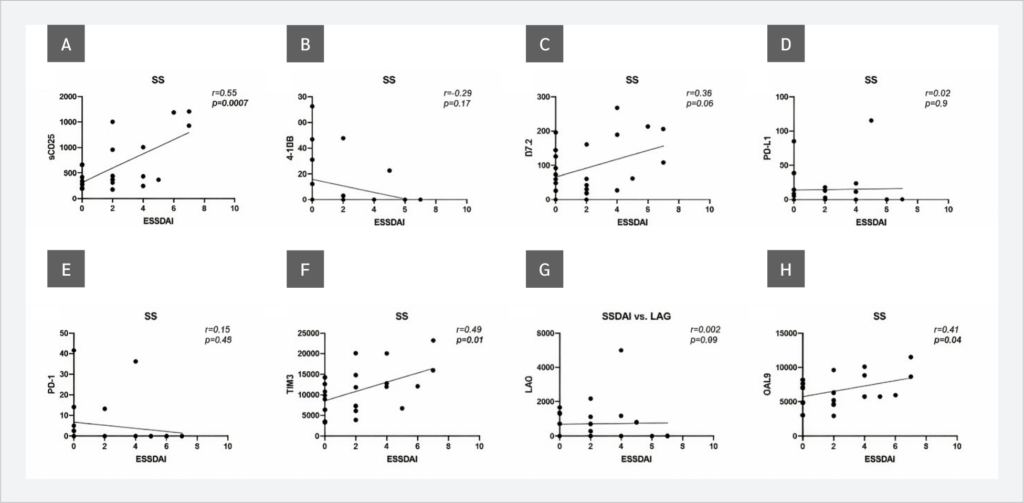
Compared with healthy controls, SLE patients had significantly elevated levels of soluble TIM-3 (sTIM-3) (p<0.0001), sLAG-3 (p<0.0001), and Gal-9 (p<0.001). A strong positive correlation was observed between sGal-9 and sTIM-3 levels (r=0.79, p<0.0001), and a moderate correlation was observed between sGal-9 and sLAG-3 levels (r=0.55, p=0.002). In SS, sICP levels did not differ significantly from controls; however, sPD-1 correlated with sPD-L1 (r=0.56, p=0.004), and sGal-9 with sTIM-3 (r=0.54, p=0.007). ESSDAI scores in SS correlated with sCD25 (r=0.65, p=0.0007), sTIM-3 (r=0.49, p=0.01), and sGal-9 (r=0.41, p=0.04).
Conclusion:
Our findings suggest the potential use of sICPs as biomarkers in SLE and SS. However, larger studies are needed to validate the diagnostic and prognostic roles of these soluble checkpoints.
Keywords:
Systemic, lupus, erythematosus Sjögren's, syndrome soluble, immune, checkpoint, proteins flow, cytometryIntroduction
Immune checkpoint proteins (ICPs) regulate mechanisms that are critical for maintaining immune tolerance. The first ICP was discovered in the early 1990s, and over the past two decades, extensive research has underscored the importance of ICPs in regulating immune responses and preventing autoimmunity (1). To date, several ICPs have been characterized, including programmed cell death protein-1 (PD-1), cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), B- and T-cell lymphocyte attenuator (BTLA), T-cell immunoglobulin and mucin-domain-containing protein 3 (TIM-3), T-cell immunoglobulin and immunoreceptor tyrosine-based inhibitory motif (ITIM) domain (TIGIT), V-domain Ig suppressor of T-cell activation (VISTA), lymphocyte activation gene 3 (LAG-3), and CD200 (2). The role of ICPs is critical not only for maintaining self-tolerance but also for preventing autoimmune diseases (3).
Systemic lupus erythematosus (SLE) and Sjögren's syndrome (SS) are chronic autoimmune diseases with complex and often overlapping features. Systemic lupus erythematosus is a multisystem disorder with a relapsing and remitting course that primarily affects women of childbearing age, with a striking female predominance of 9:1 (4). Systemic lupus erythematosus is particularly characterized by the presence of antibodies against nuclear and cytoplasmic antigens, as well as other autoantibodies such as anti-Scl-70, anti-La, anti-Ro, anti-cardiolipin, and antiphospholipid antibodies (5), suggesting an association between SLE and other autoimmune diseases. Similarly, SS is a systemic autoimmune disease of unclear etiology that primarily targets the salivary and lacrimal glands, leading to xerostomia and xerophthalmia (6). Sjögren's syndrome is a systemic disease with multisystem involvement and diverse clinical manifestations, commonly associated with fatigue, mood disorders, and reduced physical performance that markedly impair quality of life (7). It is classified as primary or secondary when occurring alone or in association with other autoimmune diseases, and its health-related quality-of-life burden is comparable to that of SLE and rheumatoid arthritis (RA) (8). Autoimmune diseases often require long-term or even lifelong medication, and some can be life-threatening and severely impair the quality of life (9).
A key feature of autoimmune diseases is the production of autoantibodies, which serve as important serologic markers for most of these diseases. However, they have limited sensitivity and specificity, requiring a more comprehensive diagnostic approach that incorporates clinical and demographic factors alongside laboratory findings (10). Recent research has highlighted the role of ICPs in regulating immune tolerance, and therapies targeting ICPs have shown promising results in the treatment of autoimmune diseases (3). Taken together, ICP expression could help to unravel disease mechanisms and identify new therapeutic targets. In the context of the association between soluble ICPs (sICPs) and autoimmune pathogenesis, this study aimed to assess the levels of 10 sICPs in serum samples from patients with SLE and SS compared with healthy controls, and to evaluate correlations among sICP levels to assess their potential as biomarkers.
Materials and Methods
Sample Collection
Inclusion criteria for patients were age ≥18 years and a diagnosis of either SLE (n=27) or SS (n=23) for this cross-sectional study. All participants were patients of the Rheumatology Department of Yeditepe University Hospital. A control group consisting of age-matched individuals without any evidence of autoimmune disease (n=23, mean age=53.9 ± 9.8; 20 [87.0%] female and 3 [13.0%] male) was also included. Ethical approval was granted by the Yeditepe University Clinical Research Ethics Committee with approval number 1631 dated July 7, 2022, and informed consent was obtained from all participants.
Disease activity was assessed using the SLE Disease Activity Index (SLEDAI) for SLE patients and the EULAR Sjögren's Syndrome Disease Activity Index (ESSDAI) for SS patients (11,12). Participants’ age, gender, systemic involvement, and autoantibody profiles were recorded. The analysis of patients' serum autoantibodies was conducted in the HLA typing laboratory at Yeditepe University Hospital for clinical evaluation.
For the evaluation of serum soluble ICP levels, 5 mL of whole blood samples were drawn from subjects into serum tubes containing clot activator (BD Vacutainer® Serum Separation [SST™] tubes; Becton, Dickinson and Company, Franklin Lakes, NJ, USA; Catalog No: 367986), and the tubes were centrifuged at 400 × g for 20 minutes. Serum samples were aliquoted and stored at -80°C in an ultrafreezer for further analysis.
Measurement of Serum Soluble Immune Checkpoint Proteins
Serum concentrations of soluble ICPs— sCD25 (soluble interleukin-2 receptor alpha [IL-2Rα; CD25]), 4-1BB (tumor necrosis factor receptor superfamily member 9 [TNFRSF9; CD137]), B7.2 (CD86; cluster of differentiation 86), transforming growth factor beta 1 (TGF-β1; free active form), cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), programmed death-ligand 1 (PD-L1), programmed cell death protein 1 (PD-1), T-cell immunoglobulin and mucin-domain-containing protein 3 (TIM-3), lymphocyte activation gene 3 (LAG-3), and Galectin-9 (Gal-9)— were quantified using the LEGENDplex™ HU Immune Checkpoint Panel 1 (10-plex) (BioLegend , San Diego, CA, USA; Catalog No: 740962). Laboratory analyses were conducted at the HLA Typing Laboratory of Yeditepe University Hospital.
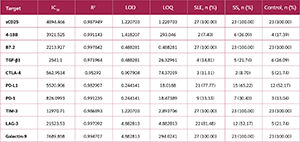
For this purpose, aliquoted serum samples stored at -80°C were thawed immediately, diluted, and the assay was performed according to the manufacturer’s instructions. Samples were acquired using a Beckman Coulter DxFLEX flow cytometry system (Beckman Coulter, Brea, CA, USA). Data were analyzed using the LEGENDplex Data Analysis Software Suite (BioLegend, San Diego, CA, USA). Performance data and patient positivity for the respective analytes are presented in Table 1.
Statistical Analysis
All statistical analyses were performed using GraphPad Prism software version 8 (GraphPad Software, San Diego, CA, USA). The Shapiro-Wilk test was used to assess the normality of the data. Comparisons between groups were performed using one-way analysis of variance (ANOVA) for parametric data or the Kruskal-Wallis test for non-parametric data. Correlations between sICPs were assessed using Pearson’s correlation for normally distributed data and Spearman’s rank correlation for non-normally distributed data. P values <0.05 were considered statistically significant.
Results
Patients’ Demographic Characteristics
A total of 27 patients with SLE were enrolled in this study, of whom 26 (96.2%) were female, and 1 (3.7%) was male. The mean age of the subjects was 43.1 ± 10.9 years, and their mean SLEDAI score was 3.7 ± 3.9. Among all SLE patients, 5 (18.5%) had hematologic involvement, 4 (14.8%) had neurologic involvement, 3 (11.1%) had nephrologic involvement, and 1 (3.7%) had both hematologic and nephrologic involvement, while 14 (52.8%) patients had no systemic involvement.
Three (11.1%) patients were anti-nuclear antibody (ANA) positive, and 15 (55.6%) were ANA negative, while ANA test results were not available for 9 (33.3%) patients. Eight (29.6%) patients were anti-double-stranded DNA (anti-dsDNA) positive, whereas 17 (62.7%) were anti-dsDNA negative; results were not available for 2 (7.4%) patients.
Among SLE patients, treatment information was unavailable for 1 (3.7%) patient. One (3.7%) patient was given a combination of prednisolone and methotrexate, while another was treated with methylprednisolone and mycophenolate mofetil. Two (7.4%) patients were administered mycophenolate mofetil monotherapy, and 8 (29.6%) patients received hydroxychloroquine alone. The remaining patients were treated with hydroxychloroquine in combination with other agents, including azathioprine, methylprednisolone, mycophenolate mofetil, hydrocortisone, colchicine, and methotrexate.
Among patients with SS, 22 (95.7%) were female, and 1 (4.3%) was male. The mean age of the subjects was 58.1 ± 14.9 years, while their mean ESSDAI score was 2.2 ± 2.3. When autoantibody status was evaluated, 14 (60.9%) patients were ANA-positive, 4 (17.4%) patients were ANA-negative, and results were unavailable for 5 (21.7%) patients. Regarding anti-SSA/Ro and anti-SSB/La antibodies, 3 (13%) patients were positive, 16 (69.6%) were negative, and results were not available for 4 (17.4%) patients.
Of the 23 patients with SS included in this study, 1 (4.3%) was under observation without medication, and 1 (4.3%) received a combination of prednisolone and methotrexate. The remaining 21 (91.3%) patients were administered hydroxychloroquine. Four (17.4%) patients received prednisolone in addition to hydroxychloroquine; 2 (8.7%) received sulfasalazine; and colchicine, methotrexate, and azathioprine were administered to 2 (8.7%), 1 (4.3%), and 1 (4.3%) patient, respectively.
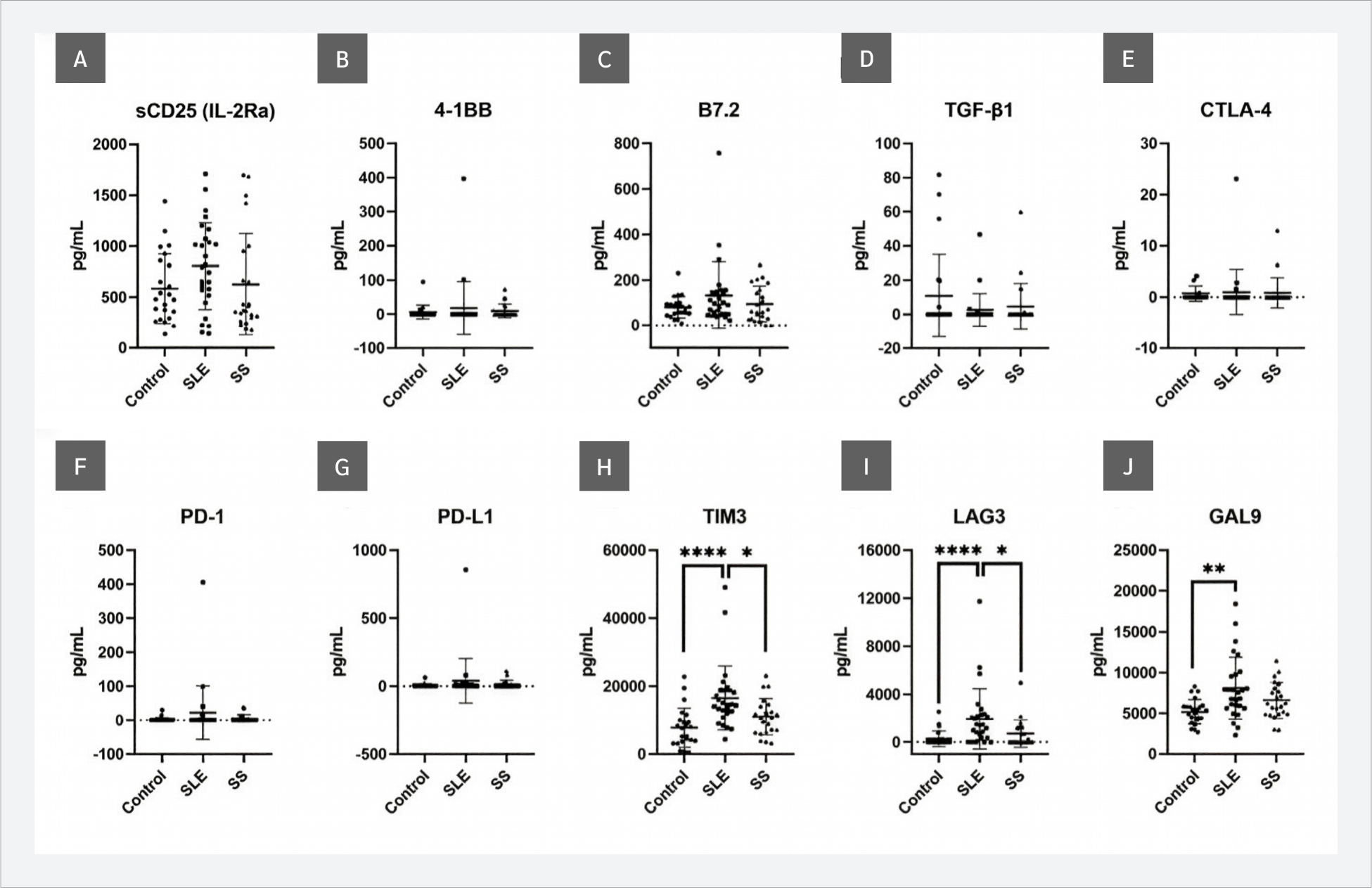
When sICP levels were evaluated, significant increases in TIM-3 (p<0.0001), LAG-3 (p<0.0001), and Gal-9 (p=0.018) were observed in SLE patients compared with the control group. No significant differences were detected in sCD25, 4-1BB, B7.2, TGF-β1, CTLA-4, PD-1, or PD-L1 levels (p>0.05). In addition to the comparison with the control group, TIM-3 and LAG-3 levels were significantly higher in the SLE group than in the SS group (p=0.0273) (Figure 1).
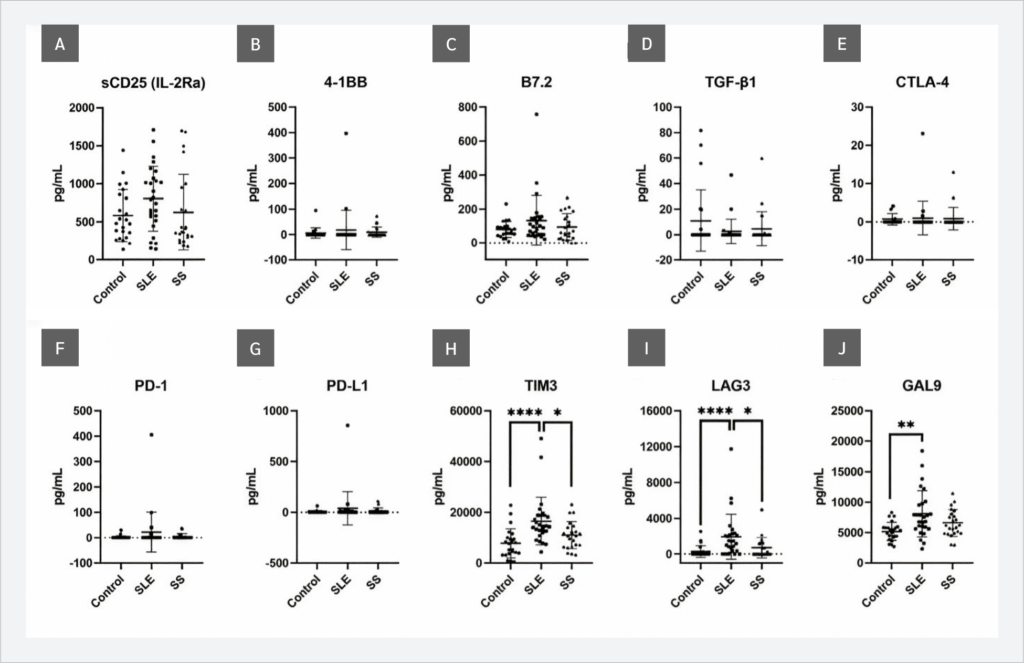
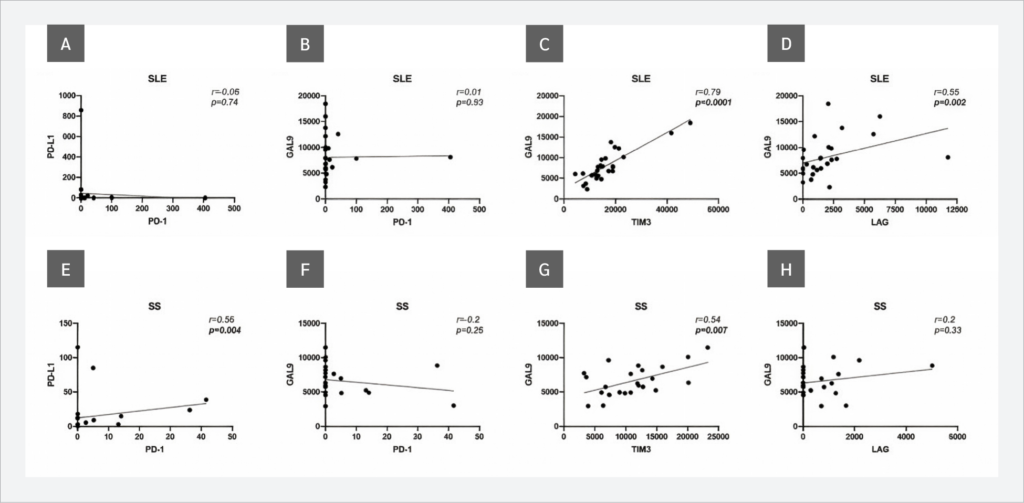
Investigations of correlations between sICP levels revealed that, in SLE patients, Gal-9 and TIM-3 showed a strong positive correlation (r=0.79, p<0.0001), and a moderate positive correlation was observed between Gal-9 and LAG-3 (r=0.55, p=0.002). Similarly, in SS patients, Gal-9 and TIM-3 levels showed a moderate positive correlation (r=0.54, p=0.007), in addition to sPD-1 and sPD-L1 (r=0.56, p=0.004) (Figure 2).
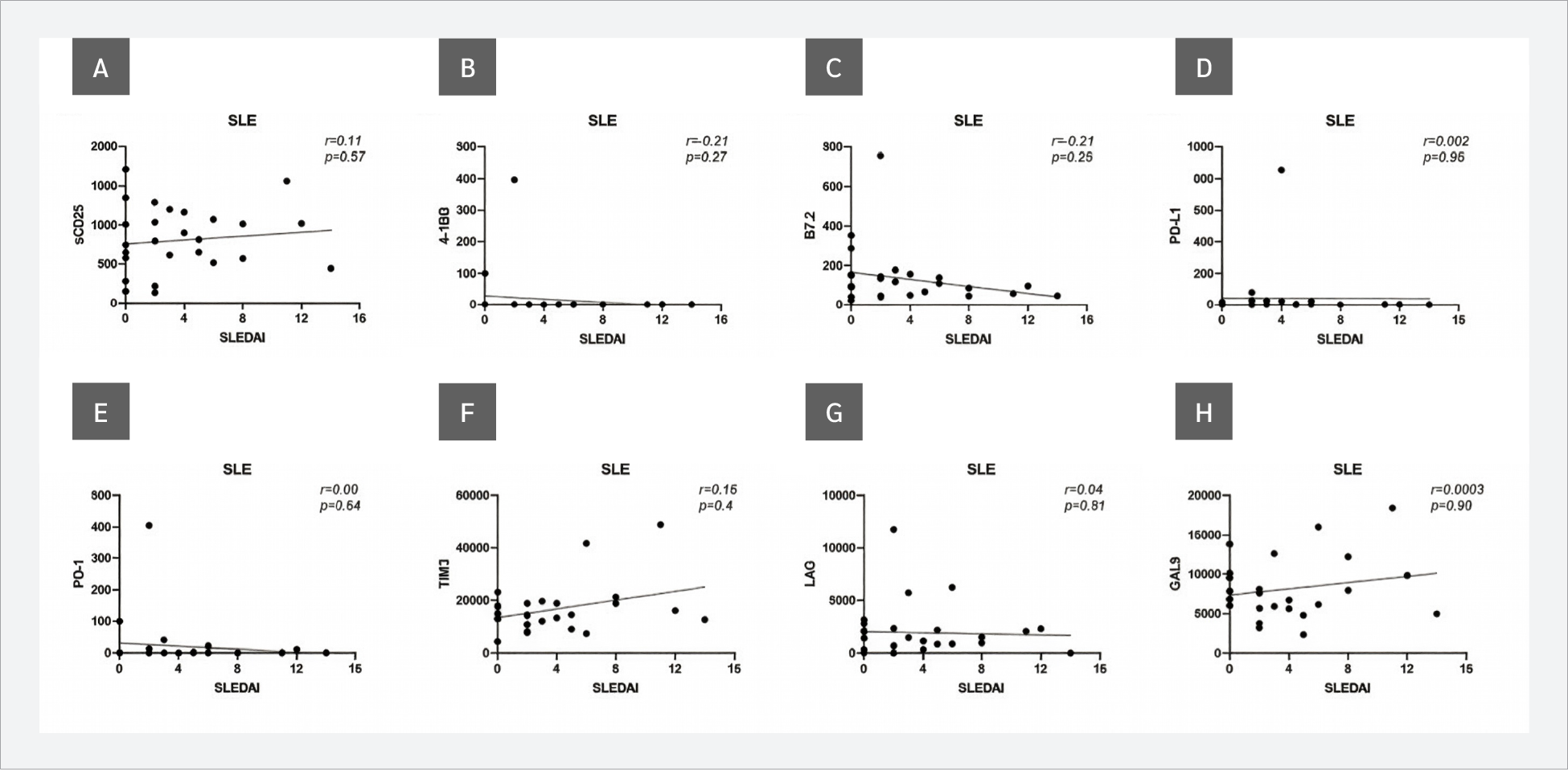
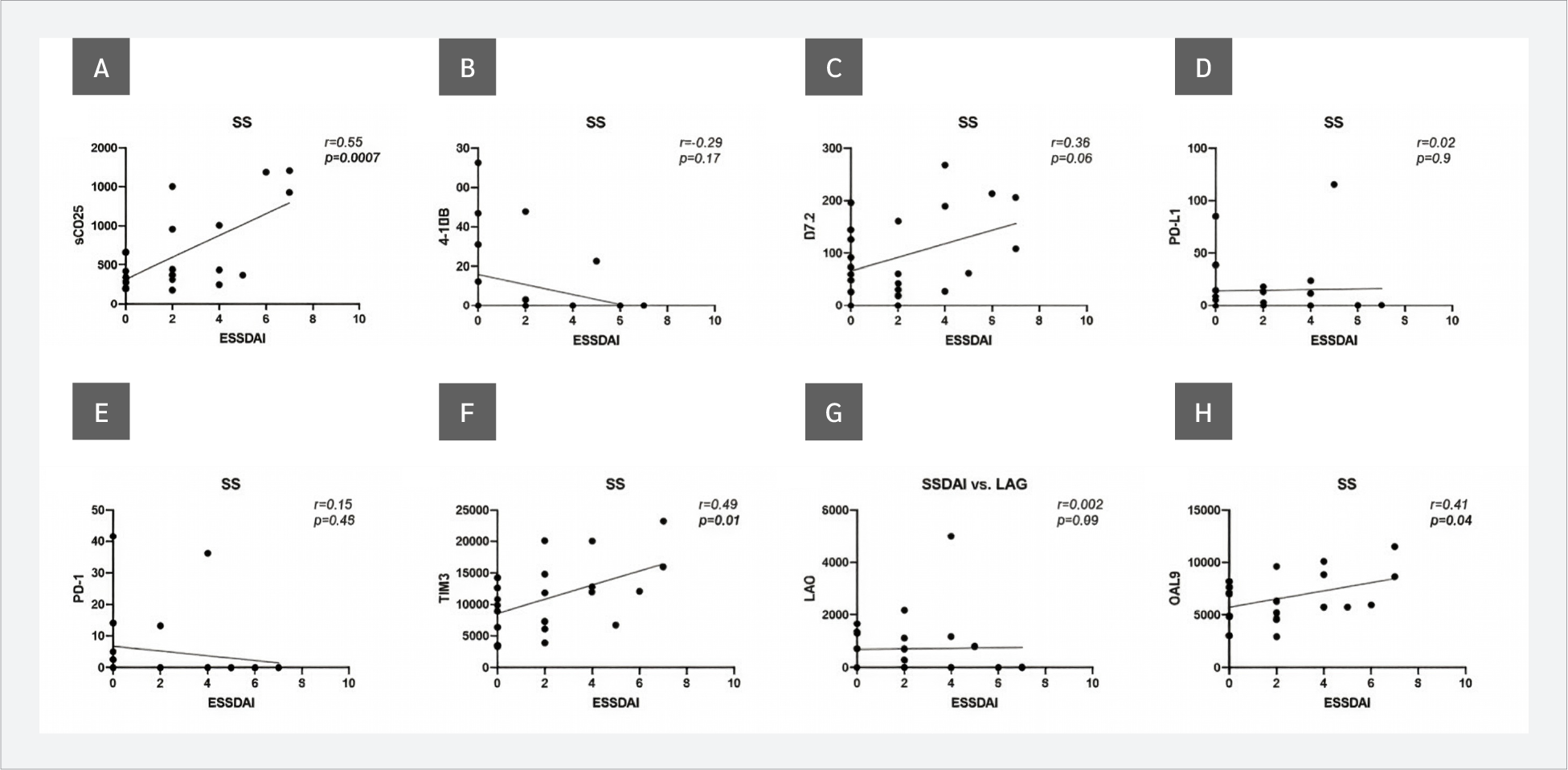
Next, the correlations between serum sICP levels and SLEDAI scores in SLE patients and ESSDAI scores in SS patients were evaluated. No correlations were observed between sICP levels and SLEDAI scores (Figure 3). However, when ESSDAI scores and sICP levels were analyzed, a strong positive correlation with sCD25 (r=0.65, p=0.0007) and weak but statistically significant positive correlations with TIM-3 (r=0.49, p=0.01) and Gal-9 (r=0.41, p=0.04) were observed. In addition, the relationship between the ESSDAI score and B7.2 levels was close to statistical significance (r=0.38, p=0.06) (Figure 4).
CTLA-4 and free active TGF-β1 were not included in the evaluation of correlations with disease activity scores because their concentrations were accurately measured in only 3 (11%) patients with SLE and in only 2 (8%) and 4 (17%) patients with SS, respectively.
Discussion
Relevant ligand-receptor pairs were evaluated when investigating correlations between sICPs. Programmed cell death protein 1 (PD-1) plays a critical role in regulating immune responses and maintaining self-tolerance by modulating T-cell activity. One of its ligands, PD-L1, is a transmembrane protein that acts as a key co-inhibitory molecule in the immune system. By binding to PD-1, PD-L1 suppresses the proliferation of PD-1-positive cells, reduces their cytokine production, and induces their apoptosis, effectively dampening the immune response (13).
Gal-9, a member of the galectin family with a tandem repeat structure, serves as a ligand for TIM-3, which is expressed on exhausted T cells (14). In addition to interacting with Gal-9, TIM-3 has been reported to bind PD-1 (15). Although no interaction between Gal-9 and LAG-3 has been reported, their co-expression has been described under certain conditions (16).
Autoimmune diseases result from a breakdown of immune tolerance and are multifactorial, with both genetic and environmental factors playing critical roles in their development (17). Although autoimmunity has a genetic component, incomplete concordance rates in monozygotic twins suggest that other factors contribute to disease onset (18). Environmental influences, such as lifestyle, infections, and diet, are supported by variations in the incidence of autoimmune diseases across different ethnic groups and geographic regions. Although autoimmune diseases rarely cause mortality, they have a significant impact on quality of life (19). Their classification remains challenging, highlighting the complexity of these conditions (20). Furthermore, the frequent coexistence of autoimmune diseases suggests shared pathogenic mechanisms, as individuals with one autoimmune disease often have an increased risk of developing another (17). To date, more than 100 autoimmune diseases have been identified, and several other conditions exhibit autoimmune-related features. It has been reported that autoimmune diseases affect approximately 5.6% to 9.4% of the global population, with their incidence and prevalence increasing significantly over the past 30 years, particularly among younger individuals (21). Because of their widespread impact, autoimmune diseases have been identified by the World Health Organization (WHO) as the third greatest threat to human health (22).
Systemic lupus erythematosus is a complex autoimmune disease that affects multiple organ systems and is characterized by the presence of autoantibodies against nuclear antigens, the accumulation of immune complexes, and chronic inflammation in key organs, including the skin, joints, and kidneys. The disease course is unpredictable, and persistent inflammation carries the risk of progressive organ damage, which may lead to serious health complications and an increased risk of premature mortality in severe cases (23). Systemic lupus erythematosus is estimated to affect more than 3.4 million people worldwide. However, determining its true global incidence and prevalence remains challenging due to variations in case definitions used in epidemiological studies and the lack of comprehensive data from many regions. The disease predominantly affects women, with a ratio of approximately 9:1 (female:male). Although the disease can occur at any age, it most commonly develops in women of childbearing age, typically between 15 and 44 years. Childhood-onset SLE, defined as disease onset before the age of 18, has been reported to be associated with a more severe disease course (23).
Sjögren's syndrome is a systemic disease characterized by multiple manifestations resulting in exocrine gland dysfunction or damage. The diagnosis of SS involves several steps, including assessment of oral and ocular dryness, detection of anti-SSA/Ro and anti-SSB/La antibodies, and performing a gland biopsy (24).
According to a meta-analysis, the incidence of SS has been estimated at 6.92 cases per 100,000 person-years, with a prevalence of 60.82 cases per 100,000 individuals (approximately 1 in 1644 people) (25). The disease is most commonly diagnosed around the ages of 40–67 years and has shown a significantly higher prevalence in women over the past 15 years (26).
The most prominent and common clinical symptom is dryness of the eyes and mouth, caused by impaired function of the salivary and lacrimal glands. In addition to dryness, patients with SS frequently suffer from chronic and debilitating symptoms, including persistent pain, dental caries, vaginal dryness, and joint pain (arthralgia), all of which contribute to a significant reduction in quality of life (27).
Immune checkpoint proteins are ligand-receptor pairs that regulate immune responses by either enhancing or suppressing immune activity (28). These molecules are primarily expressed on cells of both the adaptive immune system, particularly T cells, and the innate immune system. They play a critical role in maintaining self-tolerance and controlling the duration and intensity of immune responses in various tissues, thereby helping to prevent excessive tissue damage. By modulating effector cell activity, immune checkpoints ensure that immune responses remain balanced (28).
Stimulatory immune checkpoints include molecules such as CD137, CD137L, OX40, OX40L, CD28, CD80, B7.2, inducible T cell costimulator (ICOS), B7-related protein 1 (B7RP1), CD27 and CD70 (29). These molecules promote T cell activation and proliferation. In contrast, inhibitory checkpoints play a role in dampening immune responses to prevent overactivation and autoimmunity (30). Key inhibitory molecules include PD-1 and its ligands PD-L1 and PD-L2, as well as cytotoxic T-lymphocyte-associated protein 4 (CTLA4), CD80, B7.2, TIM-3, Gal-9, LAG-3, B and T lymphocyte attenuator (BTLA), herpesvirus entry mediator (HVEM), TIGIT, B7-H3, and B7-H4(31). Together, these stimulatory and inhibitory pathways play an essential role in fine-tuning immune responses (32). The roles of ICPs, both in membrane-bound and soluble forms, are now well documented in cancer (2,33–35). However, their involvement in autoimmune diseases remains less well understood.
TIM-3 is a member of the TIM family of immunoregulatory proteins (36). Its soluble form, which can be released from the cell surface through cleavage by membrane-associated proteases, suggests that TIM-3 also functions as a cell-free ligand (37). Through interaction with its ligand Galectin-9, TIM-3 plays an important role in the onset and progression of chronic autoimmune diseases and has therefore been proposed as a potential target for novel therapeutic strategies, including in SLE. Song et al. (38) demonstrated that increased TIM-3 expression on peripheral T-lymphocyte subsets in SLE patients is associated with higher disease activity. Similarly, Zhao et al. (39) reported that, in its soluble form, plasma TIM-3 levels positively correlate with anti-dsDNA positivity and SLEDAI scores, suggesting that soluble TIM-3 (sTIM-3) may serve as a biomarker of disease activity in SLE. In line with these studies, TIM-3 levels were significantly higher in SLE patients compared with both the control and SS groups in our study.
In addition to TIM-3, the significant increase in its ligand Gal-9 compared with the control group, as well as the strong positive correlation between sTIM-3 and Gal-9 in SLE patients, further supports activation of this pathway in SLE (40-42). On the other hand, no relationship was observed between SLEDAI scores and TIM-3 levels in our study. This finding might be attributed to the fact that the patients included in our study were not newly diagnosed and had already been receiving treatment, which may have reduced disease activity scores, as reported in previous studies (43,44). Besides its interaction with TIM-3, Gal-9 has also been shown to bind PD-1 (12), although no correlation between these two ICPs was observed in SLE patients in our study. Interestingly, serum LAG-3 levels were also significantly higher in SLE patients. LAG-3 has recently been recognized as a promising target for inhibitory drug development. Experimental studies have shown that the absence of LAG-3 does not induce autoimmune responses in vivo and that modulation of LAG-3-expressing T cells may reduce autoimmune symptoms (45). Therefore, LAG-3 has been proposed as a potential therapeutic target in SLE (45,46) and our findings further support this hypothesis. Moreover, a positive correlation between Gal-9 and LAG-3 levels was observed, which is consistent with previous reports (41).
Compared with SLE, relatively few studies have investigated the role of ICPs in SS. In its membrane-bound form, the role of PD-1 in SS remains controversial. Kobayashi et al. (47) reported increased PD-1 expression on infiltrating lymphocytes in the salivary glands of SS patients, whereas Zhai et al. (48) identified PD-1-expressing CD8+CXCR5+ cells in patients with primary SS, particularly in those with lung involvement. However, immunotherapies targeting the PD-1/PD-L1 axis have been reported to induce SS (49-51). Similar to PD-1, increased PD-L1 and CTLA-4 expression in the salivary glands of SS patients has also been reported (52). In our study, although serum levels of sPD-1 and sPD-L1 did not differ significantly between the SS and control groups, a moderate positive correlation between these two ICPs was observed which may support their involvement in SS. In contrast, a recent study suggested that TIM-3 expression is downregulated in T cells of SS patients (53). As a ligand of TIM-3, Gal-9 has been proposed as a biomarker for several autoimmune diseases, including SS (54). In our study, Gal-9 levels did not differ significantly between SS patients and controls, although a moderate positive correlation between Gal-9 and sTIM-3 was observed. In a study by van den Hoogen et al. (55), Gal-9 levels were reported to be elevated in primary Sjögren’s disease compared with non-Sjögren sicca and showed a positive correlation with ESSDAI scores. The ESSDAI was introduced by EULAR to standardize the assessment of disease activity in SS and is widely used in both clinical trials and routine practice (56). In our study, a strong correlation between ESSDAI scores and serum sCD25 levels was detected. This finding is consistent with previous studies suggesting the involvement of sCD25 in autoimmune diseases, including primary SS (57, 58). In addition, weak but statistically significant positive correlations were observed between ESSDAI scores and both Gal-9 and sTIM-3. Nevertheless, the absence of significant increases in sICP levels between SS patients and healthy controls in our study highlights the need for studies including larger patient cohorts.
Overall, the significant increase in TIM-3, LAG-3, and Gal-9 levels in the SLE group is consistent with previous reports. However, no significant correlations were observed between these markers and disease activity parameters. In contrast, although no significant differences in sICP levels were detected between the SS group and healthy controls, sCD25, TIM-3, and Gal-9 plasma levels showed positive correlations with disease activity scores. Considering the relatively low mean disease activity in our cohort and the fact that the participants were receiving treatment at the time of sampling, these correlations should be interpreted cautiously and require further validation in treatment-naïve patients and larger study populations.
Conclusion
The results of our study are consistent with previous reports and suggest that sICPs may serve as potential biomarkers in SLE and SS. Among the 10 sICPs evaluated, TIM-3, Gal-9 and LAG-3 levels were significantly higher in SLE patients, and Gal-9 levels showed positive correlations with TIM-3 and LAG-3. However, no association was observed between SLEDAI scores and sICP levels. In SS patients, sICP levels were not significantly altered compared with those of controls and SLE patients, although PD-1/PD-L1 and TIM-3/Gal-9 levels showed a positive correlation. In addition, ESSDAI scores were positively correlated with TIM-3 and Gal-9 levels.
Our findings indicate that even in treated SLE patients, sICP levels remain significantly higher than those observed in healthy controls. Further studies including treatment-naïve patients and individuals with higher disease activity are needed to confirm the potential of sICPs as biomarkers for SLE and SS and to clarify their relationship with disease severity.
Ethical Approval
The study was approved by the Yeditepe University Clinical Research Ethics Committee on July 07, 2022, with the decision number 1631.
Informed Consent
Informed consent was obtained from all participants
Peer-review
Externally peer-reviewed
Author Contributions
.K., G.Y.D.; Fundings – E.G., G.Y.D.; Materials – E.G., B.A., BMK, G.Y.D.; Data Collection and/or Processing – E.G., B.A., BMK, G.Y.D.; Analysis and/or Interpretation – B.A., M.B.K., G.Y.D.; Literature Review – E.G., B.A.; Writer – E.G., B.A., G.Y.D.; Critical Reviews – M.B.K., G.Y.D.
Conflict of Interest
The author declares no conflict of interest.
Financial Disclosure
This study was supported by the Scientific and Technological Research Council of Türkiye (TÜBİTAK) under the 2209-A Research Funding Program for University Students (2021).
AI Statement
Anthropic's Claude AI was used to check the grammar of selected paragraphs.
Scientific Presentation
This study was presented as a poster at the 7th European Congress of Immunology (ECI), held in Dublin, Ireland, from September 1 to 4, 2024.
References
Linsley PS, Greene JL, Brady W, Bajorath J, Ledbetter JA, Peach R. Human B7-1 (CD80) and B7-2 (CD86) bind with similar avidities but distinct kinetics to CD28 and CTLA-4 receptors. Immunity. 1994;1(9):793–801. Erratum in: Immunity 1995;2(2):following 203. [CrossRef]
Gaikwad S, Agrawal MY, Kaushik I, Ramachandran S, Srivastava SK. Immune checkpoint proteins: Signaling mechanisms and molecular interactions in cancer immunotherapy. Semin Cancer Biol. 2022;86(Pt 3):137–50. [CrossRef]
Zhai Y, Moosavi R, Chen M. Immune checkpoints, a novel class of therapeutic targets for autoimmune diseases. Front Immunol. 2021;12:645699. [CrossRef]
Ameer MA, Chaudhry H, Mushtaq J, Khan OS, Babar M, Hashim T, et al. An overview of systemic lupus erythematosus (SLE) pathogenesis, classification, and management. Cureus. 2022;14(10):e30330. [CrossRef]
Didier K, Bolko L, Giusti D, Toquet S, Robbins A, Antonicelli F, et al. Autoantibodies associated with connective tissue diseases: what meaning for clinicians? Front Immunol. 2018;9:541. [CrossRef]
Negrini S, Emmi G, Greco M, Borro M, Sardanelli F, Murdaca G, Indiveri F, Puppo F. Sjögren's syndrome: a systemic autoimmune disease. Clin Exp Med. 2022;22(1):9–25. [CrossRef]
Segal BM, Pogatchnik B, Holker E, Liu H, Sloan J, Rhodus N, et al. Primary Sjogren's syndrome: cognitive symptoms, mood, and cognitive performance. Acta Neurol Scand. 2012;125(4):272–8. [CrossRef]
McCoy SS, Bartels CM, Saldanha IJ, Bunya VY, Akpek EK, Makara MA, et al. National Sjögren's Foundation Survey: Burden of oral and systemic involvement on quality of life. J Rheumatol. 2021;48(7):1029–36. [CrossRef]
Pisetsky DS. Pathogenesis of autoimmune disease. Nat Rev Nephrol. 2023;19(8):509–24. [CrossRef]
Sciascia S, Bizzaro N, Meroni PL, Dimitrios B, Borghi MO, Bossuyt X, et al. Autoantibodies testing in autoimmunity: Diagnostic, prognostic and classification value. Autoimmun Rev. 2023;22(7):103356. [CrossRef]
Touma Z, Urowitz MB, Gladman DD. Systemic lupus erythematosus disease activity index 2000 responder index-50 website. J Rheumatol. 2013;40(5):733. [CrossRef]
Seror R, Bootsma H, Saraux A, Bowman SJ, Theander E, Brun JG, et al; EULAR Sjögren's Task Force. Defining disease activity states and clinically meaningful improvement in primary Sjögren's syndrome with EULAR primary Sjögren's syndrome disease activity (ESSDAI) and patient-reported indexes (ESSPRI). Ann Rheum Dis. 2016;75(2):382–9. [CrossRef]
Han Y, Liu D, Li L. PD-1/PD-L1 pathway: current researches in cancer. Am J Cancer Res. 2020;10(3):727–42.
Elahi S, Niki T, Hirashima M, Horton H. Galectin-9 binding to Tim-3 renders activated human CD4+ T cells less susceptible to HIV-1 infection. Blood. 2012;119(18):4192–204. [CrossRef]
Yang R, Sun L, Li CF, Wang YH, Yao J, Li H, et al. Galectin-9 interacts with PD-1 and TIM-3 to regulate T cell death and is a target for cancer immunotherapy. Nat Commun. 2021;12(1):832. [CrossRef]
Okoye I, Xu L, Motamedi M, Parashar P, Walker JW, Elahi S. Galectin-9 expression defines exhausted T cells and impaired cytotoxic NK cells in patients with virus-associated solid tumors. J Immunother Cancer. 2020;8(2):e001849. [CrossRef]
Samuels H, Malov M, Saha Detroja T, Ben Zaken K, Bloch N, Gal-Tanamy M, et al. Autoimmune disease classification based on PubMed text mining. J Clin Med. 2022;11(15):4345. [CrossRef]
Generali E, Ceribelli A, Stazi MA, Selmi C. Lessons learned from twins in autoimmune and chronic inflammatory diseases. J Autoimmun. 2017;83:51-61. [CrossRef]
Shiha MG, Chetcuti Zammit S, Elli L, Sanders DS, Sidhu R. Updates in the diagnosis and management of coeliac disease. Best Pract Res Clin Gastroenterol. 2023;64-65:101843. [CrossRef]
Casal Moura M, Merkel PA, Jayne D, Cid MC, Basu N, Hellmich B, et al. Challenges in the diagnosis, classification and prognosis of ANCA-associated vasculitis. Nat Rev Rheumatol. 2025;21(12):719–36. [CrossRef]
Ohta A, Nagai M, Nishina M, Tomimitsu H, Kohsaka H. Age at onset and gender distribution of systemic lupus erythematosus, polymyositis/dermatomyositis, and systemic sclerosis in Japan. Mod Rheumatol. 2013;23(4):759–64. [CrossRef]
Yuan L, Wang Y, Shen X, Ma F, Wang J, Yan F. Soluble form of immune checkpoints in autoimmune diseases. J Autoimmun. 2024;147:103278. [CrossRef]
Hoi A, Igel T, Mok CC, Arnaud L. Systemic lupus erythematosus. Lancet. 2024;403(10441):2326-2338. Erratum in: Lancet. 2024;403(10441):2292. [CrossRef]
Zhan Q, Zhang J, Lin Y, Chen W, Fan X, Zhang D. Pathogenesis and treatment of Sjogren's syndrome: Review and update. Front Immunol. 2023;14:1127417. [CrossRef]
Qin B, Wang J, Yang Z, Yang M, Ma N, Huang F, Zhong R. Epidemiology of primary Sjögren's syndrome: a systematic review and meta-analysis. Ann Rheum Dis. 2015 Nov;74(11):1983–9. [CrossRef]
Thurtle E, Grosjean A, Steenackers M, Strege K, Barcelos G, Goswami P. Epidemiology of Sjögren's: a systematic literature review. Rheumatol Ther. 2024;11(1):1–17. [CrossRef]
Cornec D, Devauchelle-Pensec V, Mariette X, Jousse-Joulin S, Berthelot JM, Perdriger A, et al. Severe health-related quality of life impairment in active primary Sjögren's syndrome and patient-reported outcomes: data from a large therapeutic trial. Arthritis Care Res (Hoboken). 2017;69(4):528–35. [CrossRef]
Zhang Y, Zheng J. Functions of immune checkpoint molecules beyond immune evasion. Adv Exp Med Biol. 2020;1248:201–26. [CrossRef]
Marin-Acevedo JA, Kimbrough EO, Manochakian R, Zhao Y, Lou Y. Immunotherapies targeting stimulatory pathways and beyond. J Hematol Oncol. 2021;14(1):78. [CrossRef]
Funes SC, Manrique de Lara A, Altamirano-Lagos MJ, Mackern-Oberti JP, Escobar-Vera J, Kalergis AM. Immune checkpoints and the regulation of tolerogenicity in dendritic cells: Implications for autoimmunity and immunotherapy. Autoimmun Rev. 2019;18(4):359–68. [CrossRef]
Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12(4):252–64. [CrossRef]
Pitts SC, Schlom J, Donahue RN. Soluble immune checkpoints: implications for cancer prognosis and response to immune checkpoint therapy and conventional therapies. J Exp Clin Cancer Res. 2024;43(1):155. [CrossRef]
He X, Xu C. Immune checkpoint signaling and cancer immunotherapy. Cell Res. 2020 Aug;30(8):660–9. [CrossRef]
Alturki NA. Review of the immune checkpoint inhibitors in the context of cancer treatment. J Clin Med. 2023;12(13):4301. [CrossRef]
Chen L, Chao Y, Li W, Wu Z, Wang Q. Soluble immune checkpoint molecules in cancer risk, outcomes prediction, and therapeutic applications. Biomark Res. 2024;12(1):95. [CrossRef]
Wolf Y, Anderson AC, Kuchroo VK. TIM3 comes of age as an inhibitory receptor. Nat Rev Immunol. 2020;20(3):173–85. [CrossRef]
Gorman JV, Colgan JD. Regulation of T cell responses by the receptor molecule Tim-3. Immunol Res. 2014;59(1-3):56–65. [CrossRef]
Song LJ, Wang X, Wang XP, Li D, Ding F, Liu HX, et al. Increased Tim-3 expression on peripheral T lymphocyte subsets and association with higher disease activity in systemic lupus erythematosus. Diagn Pathol. 2015;10:71. [CrossRef]
Zhao D, Li C, Yang X, Yan W, Zhang Y. Elevated soluble Tim-3 correlates with disease activity of systemic lupus erythematosus. Autoimmunity. 2021;54(2):97–103. [CrossRef]
Zeggar S, Watanabe KS, Teshigawara S, Hiramatsu S, Katsuyama T, Katsuyama E, et al. Role of Lgals9 deficiency in attenuating nephritis and arthritis in BALB/c mice in a pristane-induced lupus model. Arthritis Rheumatol. 2018;70(7):1089–101. [CrossRef]
Yuksel K, Sag E, Demir S, Özdel S, Kaya UA, Atalay E, et al. Plasma checkpoint protein levels and galectin-9 in juvenile systemic lupus erythematosus. Lupus. 2021;30(6):998–1004. [CrossRef]
Matsuoka N, Fujita Y, Temmoku J, Furuya MY, Asano T, Sato S, et al. Galectin-9 as a biomarker for disease activity in systemic lupus erythematosus. PLoS One. 2020;15(1):e0227069. [CrossRef]
Li M, Liang J, Pan W, Liu L, Wu M, Ding F, et al. Predictors of improvement in disease activity in first hospitalized patients with systemic lupus erythematosus: a multicenter retrospective study of a Chinese cohort. Clin Rheumatol. 2022;41(11):3355–62. [CrossRef]
Kamanamool N, Ingsathit A, Rattanasiri S, Ngamjanyaporn P, Kasitanont N, Chawanasuntorapoj R, et al. Comparison of disease activity between tacrolimus and mycophenolate mofetil in lupus nephritis: a randomized controlled trial. Lupus. 2018;27(4):647–56. [CrossRef]
Wang B, Zhang B, Wu M, Xu T. Unlocking therapeutic potential: Targeting lymphocyte activation Gene-3 (LAG-3) with fibrinogen-like protein 1 (FGL1) in systemic lupus erythematosus. J Transl Autoimmun. 2024;9:100249. [CrossRef]
Chen K, Li X, Shang Y, Chen D, Qu S, Shu J, et al. FGL1-LAG3 axis impairs IL-10-Producing regulatory T cells associated with Systemic lupus erythematosus disease activity. Heliyon. 2023;9(10):e20806. [CrossRef]
Kobayashi M, Kawano S, Hatachi S, Kurimoto C, Okazaki T, Iwai Y, et al. Enhanced expression of programmed death-1 (PD-1)/PD-L1 in salivary glands of patients with Sjögren's syndrome. J Rheumatol. 2005;32(11):2156–63.
Zhai X, Wang Y, Guo H, Liang Z, Feng M, Wu Y, Qin Y, Zhao X, Gao C, Luo J. Altered levels of circulating CD8+CXCR5+PD-1+T follicular cytotoxic cells in primary Sjögren's syndrome. Clin Rheumatol. 2022;41(6):1697–708. [CrossRef]
Caeyman A, Vandekerckhove O, Pat K, Wynants J, Weytjens K, de Wergifosse I, et al. Sjögren's syndrome caused by PD-1 inhibition in a lung cancer patient. Case Rep Oncol. 2023;16(1):1095–9. [CrossRef]
Ramos-Casals M, Maria A, Suárez-Almazor ME, Lambotte O, Fisher BA, Hernández-Molina G, et al; ICIR. Sicca/Sjögren's syndrome triggered by PD-1/PD-L1 checkpoint inhibitors. Data from the International ImmunoCancer Registry (ICIR). Clin Exp Rheumatol. 2019;37 Suppl 118(3):114–22.
Pellegrino C, D'Antonio C, Ierinò D, Onesti CE, Aschelter AM, Santini D, et al. Sjögren syndrome induced by anti PDL-1 treatment for TNBC: case report and review of literature. Front Immunol. 2024;15:1417444. [CrossRef]
An Q, Zhao J, Zhu X, Yang B, Wu Z, Su Y, et al. Exploiting the role of T cells in the pathogenesis of Sjögren's syndrome for therapeutic treatment. Front Immunol. 2022;13:995895. [CrossRef]
Sun T, Liu S, Yang G, Zhu R, Li Z, Yao G, et al. Mesenchymal stem cell transplantation alleviates Sjögren's syndrome symptoms by modulating Tim-3 expression. Int Immunopharmacol. 2022;111:109152. [CrossRef]
Moar P, Tandon R. Galectin-9 as a biomarker of disease severity. Cell Immunol. 2021;361:104287. [CrossRef]
van den Hoogen LL, van der Heijden EHM, Hillen MR, Mertens JS, Fritsch-Stork RDE, Radstake TRDJ, et al. Galectin-9 reflects the interferon signature and correlates with disease activity in systemic autoimmune diseases. Response to: 'Biomarkers: to be or not to be' by Yavuz and Rönnblom. Ann Rheum Dis. 2020;79(1):e9. [CrossRef]
Seror R, Bowman SJ, Brito-Zeron P, Theander E, Bootsma H, Tzioufas A, et al. EULAR Sjögren's syndrome disease activity index (ESSDAI): a user guide. RMD Open. 2015;1(1):e000022. [CrossRef]
Cao S, Liu X, Li Y, Yang Y, Cai X, Cong S, et al. Serum sCD25 is an indicator for rheumatoid arthritis-associated interstitial lung disease. Clin Exp Rheumatol. 2024;42(3):633–41. [CrossRef]
Chen J, Jin Y, Li C, Gan Y, Li J, Chen S, Sun X, He J, Li Z. Evaluation of soluble CD25 as a clinical and autoimmune biomarker in primary Sjögren's syndrome. Clin Exp Rheumatol. 2020;38 Suppl 126(4):142–9.
VOLUME
,
ISSUE
Correspondence
Received
Accepted
Published
Suggested Citation
DOI
License