Investigation of Immunoblot Results of Antinuclear Antibody Test Positive Patients with Systemic Autoimmune Rheumatic Diseases
Abstract
Objective:
Systemic autoimmune rheumatic diseases (SARDs) are associated with antinuclear antibodies (ANAs). This study aimed to retrospectively assess the outcomes of extractable nuclear antigen (ENA) tests in patients with rheumatologic disorders who tested positive for antinuclear antibodies.
Materials and Methods:
The study included 542 patients with a positive ANA result, no mixed pattern, and a positive immunoblot (IB) test. An ANA indirect immunofluorescence (IIF) test was performed on the same slide using the Hep-20-10/liver tissue kit (Euroimmun AG, Lübeck, Germany). Antibodies targeting ENAs were investigated using the EUROLINE ANA Profile 3 plus DFS70 (IgG) test kit (Euroimmun AG, Germany).
Results:
The homogeneous pattern (AC-1), Sm-RNP, and ribosomal P protein exhibited significant associations with systemic lupus erythematosus (SLE) (p<0.001, p=0.002, p<0.001, respectively). The speckled pattern (AC 4/5), SS-A, SS-B, and Ro52 showed a strong association with Sjögren's syndrome (p<0.001 for each). The centromere pattern (AC-3), CENP-B, and Scl-70 were significantly correlated with systemic sclerosis (p<0.001 for all). The homogeneous pattern (AC-1) and Mi-2 were significantly associated with rheumatoid arthritis (p=0.019, p=0.035, respectively). The speckled pattern (AC-4/5), Jo-1, and Mi-2 exhibited significant associations with polymyositis/dermatomyositis (p<0.001, p=0.006, p<0.001, respectively). Systemic sclerosis was substantially correlated with mixed connective tissue disease (p=0.021).
Conclusion:
Antinuclear antibody IIF testing should be used to evaluate autoimmune disorders. Extractable nuclear antigen testing is essential for identifying and confirming the presence of the antigen. Extractable nuclear antigen tests detect specific antigens, making them diagnostically and prognostically valuable in rheumatological diseases. Through rational laboratory use, these tests can enhance efficiency and provide more accurate clinical guidance.
Keywords:
Antinuclear, antibodies systemic, autoimmune, rheumatic, diseases extractable, antinuclear, antibodiesIntroduction
Autoantibodies seen in systemic autoimmune rheumatic diseases (SARDs) generally target nuclear antigens and are known as antinuclear antibodies (ANAs) (1). Antinuclear antibody-associated rheumatic diseases encompass a diverse range of conditions characterized by the presence of positive ANAs (2). The patterns identified through the indirect immunofluorescence (IIF) technique are associated with specific diseases, and anti-cell codes (ACs) have been established based on international consensus regarding ANA patterns (3).
The IIF method using HEp-2 cells is considered the gold standard for ANA screening (4). The patterns identified by IIF are associated with specific diseases. ACs have been defined in accordance with the international consensus on ANA patterns (3). Nuclear patterns include homogeneous, speckled, dense fine speckled (DFS70), nucleolar, nuclear membrane, centromere, cytoplasmic, and mitotic staining. The association between rheumatic diseases and IIF patterns is a result of autoantibodies targeting specific cellular antigens (5). Certain autoantibodies have been isolated and designated as extractable antinuclear antibodies (ENAs).
Among ENAs, the Smith (Sm) antigen is a low-molecular-weight, non-histone, acidic ribonucleoprotein. Sjögren’s syndrome-related antigen A (SS-A) is a protein involved in mRNA processing, whereas Sjögren’s syndrome-related antigen B (SS-B) is a phosphoprotein that functions as a cofactor for RNA polymerase III (6). The Scl-70 antigen is identified as DNA topoisomerase I, while Jo-1 corresponds to the enzyme histidyl-tRNA synthetase (7). Analyzing ENA responses can aid in differentiating among various forms of autoimmune connective tissue disorders (8). The presence of antibodies against the Sm antigen is specific for systemic lupus erythematosus (9), while the presence of anti-SS-A or anti-SS-B antibodies indicates Sjögren’s syndrome (7). Detection of ENA antibodies is not only diagnostically important but also holds prognostic significance. The presence of SS-A in the circulation of a pregnant woman may lead to neonatal lupus erythematosus (11) or congenital heart block in the baby, while the presence of anti-topoisomerase I (Topo-I) antibodies predicts a more severe progression of systemic sclerosis (SSc) (12). This study aimed to retrospectively assess the outcomes of extractable nuclear antigen tests in patients with rheumatologic disorders who tested positive for antinuclear antibodies.
Materials and Methods
This study retrospectively analyzed the simultaneous requests for IIF staining patterns, ANA tests, and IB tests for patients admitted to the Medical Microbiology Laboratory of Ankara Bilkent City Hospital from February 2019 to June 2023, along with their diagnoses.
The study included 542 patients with positive ANA results, no mixed pattern, and a positive IB test. Of these, 120 patients were diagnosed with systemic lupus erythematosus (SLE), 100 with Sjögren's syndrome (SS), 100 with systemic sclerosis (SSc), 100 with rheumatoid arthritis (RA), 100 with mixed connective tissue disease, and 20 with polymyositis/dermatomyositis. 90% of patients were recruited from the rheumatology department, with the remainder from internal medicine, physical therapy, and rehabilitation departments.
The ANA IIF test was performed on the same slide utilizing a kit (Euroimmun AG, Lübeck, Germany) containing Hep-20-10/liver tissues. The manufacturer recommended an initial dilution of 1:100 for screening purposes. Results were qualitatively evaluated using a EUROSTAR III plus fluorescence microscope at X40 magnification, ranging from 1+ (1:100) to 4+ (1:3200), based on the brightness observed at the screening dilution.
The EUROLINE ANA Profile 3 plus DFS70 (IgG) test kit (Euroimmun AG, Germany) was used to analyze antibodies targeting ENA. The examination identified 18 distinct antigens on the IB strips, including Sm/RNP, Sm, SS-A, SS-B, Scl-70, Jo-1, dsDNA, nucleosome, histone, ribosomal P-protein, AMA M2, Ro-52, PM/Scl, CENP-B, PCNA, Ku, Mi-2, and DFS70. All incubation and washing procedures were performed manually using the EUROBlotOne system (Euroimmun). Band intensities were evaluated with EUROLineScan software (Euroimmun). Certain autoantibodies were isolated and classified as ENA, which are commonly identified using enzyme-linked immunosorbent assay (ELISA) or IB techniques.
The study was approved by the Non-Interventional Clinical Research Ethics Committee of Ankara Bilkent City Hospital on July 12, 2023, with the decision number E2-23-4480.
Statistical Analysis
Statistical analyses were performed using IBM SPSS Statistics for Windows, version 22.0 (IBM Corp., Armonk, NY). The data were assessed for normal distribution using the Kolmogorov-Smirnov test. Numerical variables following a normal distribution were expressed as mean ± standard deviation, while non-normally distributed data were expressed as median values. Categorical data were compared using either the Chi-square test or Fisher's exact test, as appropriate. A significance level of p<0.05 was considered statistically significant.
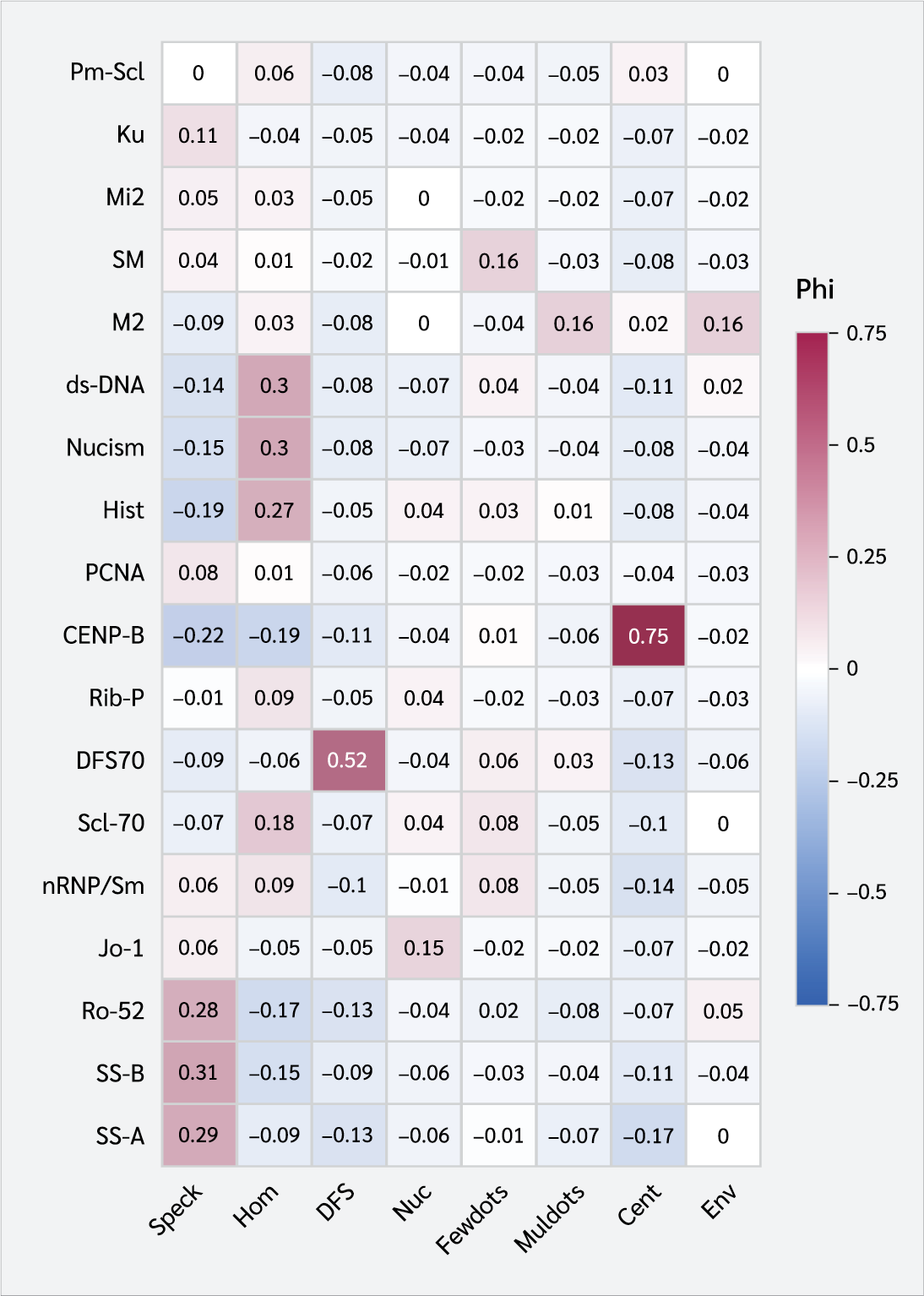
Correlation analysis and heatmap visualization were conducted using the psych and ggplot2 packages in R statistical software (version 4.5.0). Phi correlation coefficients were calculated to determine the strength and direction of associations (range: -1 to +1). Red shades indicated positive correlations (darker red denoting stronger relationships), blue indicated negative correlations, and white (φ = 0) denoted no correlation.
Results
All 100 patients diagnosed with rheumatological diseases, including SLE, SS, SSc, RA, mixed connective tissue disease, and polymyositis/dermatomyositis, in a randomly selected cohort from February 2019 to June 2023 exhibited ANA positivity, a single ANA pattern, and a positive IB test. The patients' mean age was 50.5 ± 16.5 years. Women accounted for 474 cases (87.5% of the total).
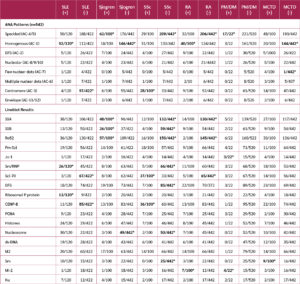
Among the ANA patterns observed, 238 were speckled (AC-4/5), 164 were homogeneous (AC-1), 61 were centromere (AC-3), 31 DFS (AC-2), 27 were nucleolar (AC-8/9/10), 8 were multiple nuclear dots (AC-6), 8 were envelope (AC-11/12), and 5 were few nuclear dots (AC-7). No cytoplasmic ANA pattern was detected.
The frequency of antigen positivity was as follows: Ro52 in 166 patients, SS-A in 144, CENP-B in 96, DFS in 92, M2 in 80, Pm-Scl in 75, Sm/RNP in 71, Scl-70 in 70, SS-B in 63, histone in 53, nucleosome in 52, dsDNA in 47, PCNA in 32, Sm in 25, ribosomal P protein in 22, Mi-2 in 19, Ku positivity in 19, and Jo-1 in 18.
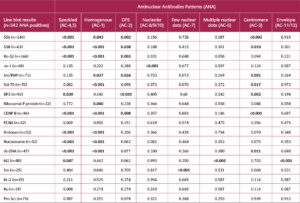
Table 1 shows the correlation between ANA patterns and IB results. SS-A, SS-B, Ro52, and Ku positivity were associated with the speckled pattern (AC-4/5) (p<0.001, p<0.001, p<0.001, p<0.001, p=0.008, respectively). Sm/RNP, Scl-70, ribosomal p protein, histone, nucleosome, dsDNA were associated with a homogeneous pattern (AC-1) (p=0.037, p<0.001, p=0.040, p<0.001, p<0.001, p<0.001, p<0.001, p<0.001, respectively). Jo-1 was associated with the nucleolar pattern (AC-8/9/10) (p<0.001). DFS positivity was associated with the DFS pattern (AC-2) (p<0.001). CENP-B positivity was associated with the centromere pattern (AC-3) (p<0.001). Sm positivity was associated with the few nuclear dots pattern (AC-7) (p<0.001). M2 positivity was associated with multiple nuclear dots (AC-7) and envelope (AC-11/12) patterns (p<0.001 for both).
Table 2 illustrates the correlations between ANA patterns, IB results, and rheumatologic diseases. The homogeneous pattern (AC-1), Sm/RNP, ribosomal p protein, histone, nucleosome, and dsDNA were strongly associated with the presence of SLE (p<0.001, p=0.002, p<0.001, p<0.001, p<0.001, and p=0.002, respectively). The centromere pattern (AC-3), Scl-70, and CENP-B were significantly associated with the absence of SLE (p=0.002, p<0.001, and p=0.005, respectively).
The speckled pattern (AC-4/5), SS-A, SS-B, and Ro52 were significantly associated with the presence of SS (p<0.001 for all). In contrast, the homogeneous pattern (AC-1) and nucleosome were significantly associated with the absence of SS (p=0.003 and p=0.013, respectively).
The centromere pattern (AC-3), CENP-B, and Scl-70 were significantly associated with the presence of SSc (p<0.001 for all). The speckled pattern (AC-4/5), SS-A, SS-B, Ro52, Sm/RNP, DFS, nucleosome, and Sm were significantly associated with the absence of SSc (p<0.001, p<0.001, p=0.008, p<0.001, p=0.008, p=0.003, p=0.004, and p=0.015, respectively).
The homogeneous pattern (AC-1) and Mi-2 were significantly associated with the presence of RA (p=0.019 and p=0.035, respectively). Conversely, the speckled pattern (AC-4/5), SS-A, Ro52, and Scl-70 were significantly associated with the absence of RA (p=0.008, p=0.002, p=0.021, and p=0.009, respectively).
The speckled pattern (AC-4/5), Jo-1, and Mi-2 were significantly associated with polymyositis/dermatomyositis (p<0.001, p=0.006, and p<0.001, respectively). Sm was significantly associated with the presence of mixed connective tissue disease (p=0.021), whereas the homogeneous pattern (AC-1) and the few nuclear dots pattern (AC-7) were significantly associated with its absence (p=0.013 and p<0.001, respectively).
According to Phi correlation analysis, the speckled pattern (AC-4/5) was correlated with SS-A, SS-B, and Ro-52. The homogeneous pattern (AC-1) correlated with dsDNA, nucleosome, histone, and Scl-70. The DFS-like pattern (AC-2) showed a strong correlation with DFS70. The nucleolar pattern (AC-8/10) correlates with Jo-1. The few nuclear dots pattern (AC-7) correlated with Sm. The multiple nuclear dots pattern (AC-6) was correlated with M2. Centromere pattern (AC-3) showed a strong correlation with CENP-B. Envelope pattern (AC-11/12) correlated with M2 (Figure 1).
Discussion
Laboratories in developing countries, including Türkiye, require a dependable, cost-effective, accurate, and specific screening test. The high cost and the need for technical expertise limit its availability in many facilities.
Antinuclear antibody detection is the first step in diagnosing SARDs (13).The IIF method using HEp-2 cells is considered the gold standard (14). If ANA results are positive, it is advisable to screen for specific anti-ENA antibodies (15). In cases of strong clinical suspicion, regardless of ANA test results, physicians may request testing for specific ENA antibodies (16).
The American College of Rheumatology (ACR) and international committees recommend HEp-2 IIF as the standard method for identifying ANA (17). To date, up to 30 distinct ANA staining patterns have been described, including both nuclear and cytoplasmic staining patterns (18).The most common patterns are homogeneous (AC-1), speckled (AC-4/5), nucleolar (AC-8/9/10), and centromere (AC-3) (19). In this study, the speckled ANA pattern was the most frequently observed. Similarly, in Trabzon province, Kaklıkkaya et al. (20) reported the speckled pattern as the most common, consistent with our findings.
The homogeneous pattern is one of the most common patterns observed in the ANA test. The antigens associated with this pattern include dsDNA, histone, and nucleosomes. In a previous study, 45% of the homogenous patterns were positive in the IB test (16). Low DFS70 titers and homogeneous/speckled patterns can be difficult to distinguish; therefore, we recommend confirming them with the IB test using the DFS70 antigen. We hypothesize that the IB findings for samples with homogeneous patterns vary due to antibody diversity, particularly in patients with SLE. The antibodies detected in these patients' dsDNA, nucleosomes, and histones include SS-A, Ro-52, SS-B, Sm, U1-RNP, and ribosomal P protein (21).
In our study, the homogenous pattern (AC-1) was associated with Sm/RNP, Scl-70, ribosomal p protein, histone, nucleosome, and dsDNA. The DFS70 staining pattern is one of the most common IIF scanning patterns (22). Isolated DFS70 antibody positivity occurs in fewer than 5.7% of systemic rheumatic diseases (22), and these antibodies are often present in the serum of healthy individuals (23). In our analysis, the DFS70 pattern was the fourth most prevalent. As expected, DFS showed no correlation with any rheumatic condition in our investigation.
The centromere pattern, characterized by its structural features, is observed in localized cutaneous systemic sclerosis and Raynaud's syndrome, and is associated with CENP-A, CENP-B, CENP-C, and CENP-F antigens (24). In a previous study, only CENP-B was identified in the IB test, with a 91% positivity rate (16). The remaining negative samples may be related to other antigens. The strong agreement between the IFF centromere pattern and CENP-B positivity suggests that IFF alone may be sufficient to identify the centromere pattern. In our analysis, CENP-B positivity showed a clear correlation with SSc.
The speckled pattern is frequently observed in conditions such as mixed connective tissue disorders, SLE, and SSc (25). It is specifically associated with the SS-A, SS-B, Topo-1, Sm, U1-SnRNP, Mi2, and Ku antigens. A recent study revealed that the majority of the IB panel results showed antibodies against SS-A, Ro-52, and SS-B (16).
Depending on the HEp-2 cell line used in the IIF assay, SS-A, Jo-1, and ribosomal P-protein antibodies may be difficult to detect (17). Due to the extremely low expression levels of these antigens in HEp-2 cells or their potential denaturation during tissue fixation, false negatives may occur. In clinically suspected cases, ENA testing should be requested even if IIF results are negative.
We analyzed the correlation between IB and ANA patterns in our cohort to identify the most cost-effective follow-up autoantibody test. In recent years, autoantibody testing for systemic rheumatic disorders has evolved significantly (26). The combined use of ANA patterns and extended IB panels has reduced both diagnostic time and the risk of misdiagnosis (27). Furthermore, the interpretation and reporting of ANA have been standardized to eliminate subjectivity (26). However, the testing, reporting, and interpretation of these autoantibodies remain challenging because of multiple factors, including the testing methodology and platform, the experience of immunologists and laboratory scientists, the variability of fluorescent microscopes, and the pre-test probability of SARDs (26). It is essential that physicians are aware of and approve the revised autoantibody reporting format.
As observed, ENA or anti-ENA profiling enables differentiation of various forms of SARDs. A previous study found that the presence of RNP autoantibodies is a useful marker in the diagnosis of mixed connective tissue disease (28). Similarly, ANA positivity along with dsDNA or Sm positivity serves as the diagnostic criterion for SLE (29). SS-A and SS-B antibodies are valuable immunological markers for identifying SS, subacute cutaneous SLE, and newborn lupus syndrome (30). Jo-1, histidyl-tRNA synthetase, has been established as an immunomarker for polydermatomyositis. Likewise, CENP-B and Scl-70 positivity support the identification of SSc (31). Our findings were consistent with these results.
Certain ANA patterns show strong associations with IB antigens. Fibrillarin, PM/Scl, RNA polymerase I, and other IB proteins are commonly found in the nucleolar pattern (32). González et al. (33) reported various nuclear patterns in sera positive for SS-A, SS-B, and Ro-52. Rodríguez-Orozco et al. (34) found that approximately 3.4% of IB-positive samples were ANA-negative. In our cohort, PM/Scl antibodies were detected in nucleolar patterns.
Accurate interpretation of these antibodies requires consideration of clinical features. Because many SARD manifestations are nonspecific and overlap, diagnosis can be challenging, and patients are sometimes initially diagnosed with multiple SARDs until proven otherwise (26). SLE is the prototype for SARD. Several autoantibodies can be identified in SLE, differing in their sensitivity and specificity (26). Certain autoantibodies, including SS-A, SS-B, and RNP, can be detected in multiple SARDs (35). In a prior cohort, they discovered that nearly all individuals with IB were positive for SLE, but only a few autoantibodies were positive in the remainder of the SARD. Some patients may have overlap syndromes, which could explain the occurrence of several patterns and autoantibodies (26). Nonetheless, applying the proposed algorithm may aid in narrowing down the final diagnosis, determining targeted therapy, and providing an adequate prognosis (35).
In a previous cohort, 62% of patients tested positive for both IB and dsDNA (26). One could argue that once a diagnosis is established through the detection of a specific autoantibody, extending the IB panel may be unnecessary. However, certain autoantibodies are associated with a more specific symptom. For example, SS-A and SS-B are linked to congenital heart block, while ribosomal p protein is linked to neuropsychiatric problems (35). Moreover, as many SARDs can coexist (26), IB testing is essential even when positive dsDNA antibodies are present. In a study conducted in Türkiye, SS-A (26.88%), SS-B (17.81%), and Sm/RNP (17.66%) were the most frequently detected ENA antibodies in ANA-positive samples (7). In our cohort, the most common IB findings were Ro52, SS-A, CENP-B, and DFS.
Specific ANA patterns and IB test positivity were each associated with some autoimmune rheumatic diseases. The identification of these immunologic markers underscores the predictive value of immunologic tests for disease diagnosis. Further prospective studies are necessary to expand the evidence base.
This study has some limitations. First, ANA tests were not performed on all patients, and ANA-negative patients were excluded, which prevented drawing conclusions regarding negative results. Second, data on cytoplasmic staining and accompanying IB results are limited. Future research should include larger patient cohorts, comprehensive IB results for all cellular patterns (nuclear, mitotic, and cytoplasmic), and detailed clinical associations. Employing an expanded IB panel, such as the myositis panel, may further clarify disease etiology and inform targeted therapies.
Conclusion
Antinuclear antibody IIF testing is recommended as a screening test for suspected autoimmune diseases. Extractable nuclear antigen testing should be employed to identify and confirm the presence of the appropriate antigen. Extractable nuclear antigen tests are both diagnostically and prognostically valuable, particularly in the diagnosis of rheumatic disorders such as systemic lupus erythematosus, Sjögren's syndrome, and systemic sclerosis. The rational implementation of these tests in laboratories can improve efficiency and inform clinical decision-making.
Ethical Approval
The study was approved by the Non-Interventional Clinical Research Ethics Committee of Ankara Bilkent City Hospital on July 12, 2023, with the decision number E2-23-4480.
Peer-review
Externally peer-reviewed
Author Contributions
Concept – E.S.; Design – E.S.; Supervision – E.S.; Fundings – E.S.; Materials – E.S.; Data Collection and/or Processing – E.S.; Analysis and/or Interpretation – E.S.; Literature Review – E.S.; Writer – E.S.; Critical Reviews – E.S.
Conflict of Interest
The author declares no conflict of interest.
Financial Disclosure
The author declared that this study has received no financial support.
References
Muro Y. Antinuclear antibodies. Autoimmunity. 2005;38(1):3-9. [CrossRef]
Littlejohn EA, Kong L, Wang L, Somers EC. Longitudinal antinuclear antibody titers in systemic lupus erythematosus and other rheumatic diseases. Front Med (Lausanne). 2024;11:1441221. [CrossRef]
Damoiseaux J, Andrade LEC, Carballo OG, Conrad K, Francescantonio PLC, Fritzler MJ, et al. Clinical relevance of HEp-2 indirect immunofluorescent patterns: the International Consensus on ANA patterns (ICAP) perspective. Ann Rheum Dis. 2019;78(7):879-89. [CrossRef]
Buchner C, Bryant C, Eslami A, Lakos G. Anti-nuclear antibody screening using HEp-2 cells. J Vis Exp. 2014;(88):e51211. [CrossRef]
Tešija Kuna A, Đerek L, Drvar V, Kozmar A, Gugo K. Assessment of antinuclear antibodies (ANA): National recommendations on behalf of the Croatian society of medical biochemistry and laboratory medicine. Biochem Med (Zagreb). 2021;31(2):020502. Erratum in: Biochem Med (Zagreb). 2022;32(1):011201. [CrossRef]
Liang C, Xiong K, Szulwach KE, Zhang Y, Wang Z, Peng J, et al. Sjogren syndrome antigen B (SSB)/La promotes global microRNA expression by binding microRNA precursors through stem-loop recognition. J Biol Chem. 2013;288(1):723-36. [CrossRef]
Gür Vural D, Tanrıverdi Çaycı Y, Bıyık İ, Bilgin K, Birinci A. Evaluation of immunoblotting test results in patients with positive antinuclear antibodies. Turk Hij Den Biyol Derg. 2021;78(4):443-50. [CrossRef]
Ferrara CA, La Rocca G, Ielo G, Libra A, Sambataro G. Towards early diagnosis of mixed connective tissue disease: Updated perspectives. Immunotargets Ther. 2023;12:79-89. [CrossRef]
van Beers JJBC, Schreurs MWJ. Anti-Sm antibodies in the classification criteria of systemic lupus erythematosus. J Transl Autoimmun. 2022;5:100155. [CrossRef]
Mariette X. Sjögren syndrome: A forgotten or an overdiagnosed entity? J Rheumatol. 2023:jrheum.2023-0964. [CrossRef]
Fredi M, Argolini LM, Angeli F, Trespidi L, Ramoni V, Zatti S, et al. Anti-SSA/Ro positivity and congenital heart block: obstetric and foetal outcome in a cohort of anti-SSA/Ro positive pregnant patients with and without autoimmune diseases. Clin Exp Rheumatol. 2023;41(3):685-93. [CrossRef]
Senécal JL, Hoa S, Yang R, Koenig M. Pathogenic roles of autoantibodies in systemic sclerosis: Current understandings in pathogenesis. J Scleroderma Relat Disord. 2020;5(2):103-29. [CrossRef]
Satoh M, Vázquez-Del Mercado M, Chan EK. Clinical interpretation of antinuclear antibody tests in systemic rheumatic diseases. Mod Rheumatol. 2009;19(3):219-28.[CrossRef]
Cinquanta L, Bizzaro N, Pesce G. Standardization and quality assessment under the perspective of automated computer-assisted HEp-2 immunofluorescence assay systems. Front Immunol. 2021;12:638863. [CrossRef]
Infantino M, Carbone T, Brusca I, Alessio MG, Previtali G, Platzgummer S, et al; Study Group on Autoimmune Diseases of the Italian Society of Clinical Pathology and Laboratory Medicine. Current technologies for anti-ENA antibody detection: State-of-the-art of diagnostic immunoassays. J Immunol Methods. 2022;507:113297. [CrossRef]
Togay A, Yılmaz N. Comparison of the results of indirect immunofluorescence and immunoblot in the detection of anti-nuclear antibodies. Anatol J Gen Med Res. 2023;33(1):136-41. [CrossRef]
Irure-Ventura J, López-Hoyos M. The past, present, and future in antinuclear antibodies (ANA). Diagnostics (Basel). 2022;12(3):647. [CrossRef]
Chan EK, Damoiseaux J, Carballo OG, Conrad K, de Melo Cruvinel W, Francescantonio PL, et al. Report of the First International Consensus on Standardized Nomenclature of Antinuclear Antibody HEp-2 Cell Patterns 2014-2015. Front Immunol. 2015;6:412.[CrossRef]
Damoiseaux JG, Tervaert JW. From ANA to ENA: how to proceed? Autoimmun Rev. 2006;5(1):10-7. [CrossRef]
Kaklıkkaya N, Akıneden A, Topbaş M, Aydın F. Determination of anti-nuclear antibody seroprevalence in adult age groups in trabzon province. Balkan Med J. 2013;30(3):343-4. [CrossRef]
Alsubki R, Tabassum H, Alfawaz H, Alaqil R, Aljaser F, Ansar S, et al. Association between antinuclear antibodies (ANA) patterns and extractable nuclear antigens (ENA) in HEp-2 cells in patients with autoimmune diseases in Riyadh, Saudi Arabia. Intractable Rare Dis Res. 2020;9(2):89-94. [CrossRef]
Cheng CF, Shih MC, Lan TY, Li KJ. Anti-DFS70 Antibodies for differentiating systemic autoimmune rheumatic disease in patients with positive ANA tests: a systematic review and meta-analysis. Diagnostics (Basel). 2021;11(9):1592. [CrossRef]
Mariz HA, Sato EI, Barbosa SH, Rodrigues SH, Dellavance A, Andrade LE. Pattern on the antinuclear antibody-HEp-2 test is a critical parameter for discriminating antinuclear antibody-positive healthy individuals and patients with autoimmune rheumatic diseases. Arthritis Rheum. 2011;63(1):191-200. Erratum in: Arthritis Rheum. 2011;63(5):1468. [CrossRef]
Kayser C, Fritzler MJ. Autoantibodies in systemic sclerosis: unanswered questions. Front Immunol. 2015;6:167. [CrossRef]
Klotz W, Herold M. Commentary: Strong association of the myriad discrete speckled nuclear pattern with anti-SS-A/Ro60 antibodies: Consensus experience of four international expert centers. Front Immunol. 2022;13:840960. [CrossRef]
Anis S, Fatima A, Abdul Jabbar S, Arain T. ANA-specific antibodies, ANA patterns, anti-ds-DNA results, and clinical diagnosis: a laboratory and clinical audit. Immunol Res. 2023;71(2):267-75. [CrossRef]
Van Hoovels L, Broeders S, Chan EKL, Andrade L, de Melo Cruvinel W, Damoiseaux J, et al. Current laboratory and clinical practices in reporting and interpreting anti-nuclear antibody indirect immunofluorescence (ANA IIF) patterns: results of an international survey. Auto Immun Highlights. 2020;11(1):17. [CrossRef]
Tani C, Carli L, Vagnani S, Talarico R, Baldini C, Mosca M, et al. The diagnosis and classification of mixed connective tissue disease. J Autoimmun. 2014;48-49:46-9. [CrossRef]
Irure-Ventura J, López-Hoyos M. Disease criteria of systemic lupus erythematosus (SLE); the potential role of non-criteria autoantibodies. J Transl Autoimmun. 2022;5:100143. [CrossRef]
Veenbergen S, Kozmar A, van Daele PLA, Schreurs MWJ. Autoantibodies in Sjögren's syndrome and its classification criteria. J Transl Autoimmun. 2021;5:100138. [CrossRef]
van Oostveen WM, Huizinga TWJ, Fehres CM. Pathogenic role of anti-nuclear autoantibodies in systemic sclerosis: Insights from other rheumatic diseases. Immunol Rev. 2024;328(1):265-82. [CrossRef]
Zhang X, Li W, Sun S, Liu Y. Advances in the structure and function of the nucleolar protein fibrillarin. Front Cell Dev Biol. 2024;12:1494631. [CrossRef]
González DA, Rodríguez CC, Armas LM, Varela AR, Rodríguez IM, Duarte MT, et al. Anti-ENA profiles related with anti-SS-A/Ro. The detection of Ro52 and Ro60 according to the presence of SS-B/La, and ANA pattern and titer. Immunol Lett. 2014;161(1):6-12. [CrossRef]
Rodríguez-Orozco AR, Béjar-Lozano C, Cortés-Rojo C. Antibodies to extractable nuclear antigens are detectable in a considerable number of sera that test negative for antinuclear antibodies. Arch Pathol Lab Med. 2022;146(2):143-4. [CrossRef]
Gryka-Marton M, Szukiewicz D, Teliga-Czajkowska J, Olesinska M. An overview of neonatal lupus with anti-ro characteristics. Int J Mol Sci. 2021;22(17):9281. [CrossRef]
VOLUME
,
ISSUE
Correspondence
Received
Accepted
Published
Suggested Citation
DOI
License