Efficacy of the Thymus Polypeptide Fraction Biomodulina T in Children with Thymic Hypoplasia and Recurrent Infections
Abstract
Objective:
Thymic hypoplasia (TH) in children is a known cause of recurrent infections, often indicative of immunodeficiency. This study aimed to evaluate the efficacy of Biomodulina T, a thymic polypeptide fraction, in pediatric patients with TH, with or without associated cellular immunodeficiency.
Materials and Methods:
A non-controlled, phase III clinical trial was conducted among children aged 1–5 years (=60) and registered in the Cuban Public Registry of Clinical Trials/ International Clinical Trials Registry Platform (ID code RPCEC00000247). Patients were divided into two groups: Group I (n=44), with TH without cellular immunodeficiency; and Group II (n=16), with TH and cellular immunodeficiency. Biomodulina T was administered intramuscularly (IM) in two 4-week cycles, separated by a 4-week rest period. Patients who had not achieved normal thymic size by week 16 received a third, 8-week cycle.
Results:
Both groups showed significant increases in thymic size from baseline, with no significant difference between them. The mean increase was 67% (95% CI 61–73%), and 86.5% of patients completed treatment with thymic size within the normal range. Bacterial infections decreased by 91.5%, and viral infections by 80.7%, accompanied by a reduction in antibiotic use. In patients with cellular immunodeficiency, Biomodulina T significantly increased in CD4+ T lymphocytes (p), while no significant changes were observed in CD8+ T cells, CD19+ B cells, and CD56+ natural killer (NK) cells. Serum immunoglobulin A (IgA) levels also increased significantly. Overall, 82.7% of patients were classified as improved, showing both normalization of the thymic size and a 50% reduction in infections.
Conclusion:
Biomodulina T was clinically effective in the treatment of TH in children, regardless of the presence of cellular immunodeficiency.
Keywords:
Thymic, hypoplasia cellular, immunodeficiency infectionsIntroduction
The thymus is a primary lymphoid organ essential for the development and maturation of immunocompetent T cells. Its size decreases progressively throughout life, with involution typically beginning after puberty (1,2). Both genetic and environmental factors regulate thymic development. Thymic hypoplasia (TH) and aplasia refer to a small or absent thymus, particularly in small children. These conditions are most commonly associated with DiGeorge Syndrome and other congenital T-cell disorders (3,4). Additionally, viral and bacterial infections can cause thymic atrophy or impair its regenerative capacity (1,2).
Although TH is not highly prevalent, it poses a substantial burden on affected children, their families, and healthcare systems. It is associated with increased risk of morbidity and mortality, high treatment costs, and excessive use of antibiotics (5). Currently, no validated treatment exists specifically for TH. However, thymic extracts have shown promising clinical results. They influence the T cell development, maturation, and cytotoxic function, as well as enhance phagocytic and cytotoxic activity (6).
Biomodulina T (BIOCEN, Cuba) is an injectable bovine thymic extract registered in Cuba as an immunomodulatory biological drug, containing low-molecular-weight polypeptides. It is indicated for conditions characterized by cellular immunological dysfunction, such as recurrent infections in the elderly. Biomodulina T stimulates lymphoblastoid mitosis and influences the maturation and differentiation of T lymphocytes (7,8). Recent studies in older adults demonstrated increases in naïve CD4+ and CD8+ T cells, the CD4+/CD8+ ratio, as well as B and natural killer (NK) cells (9-12).
Between 1999 and 2007, Mollineda et al. (13), Christian et al. (14,15), and Rabassa et al. (16) were the first to report the use of Biomodulina T for treating TH in pediatric patients with recurrent infections. These studies showed favorable clinical outcomes, although immunological responses were not evaluated.
The objective of the present study was to evaluate the efficacy of Biomodulina T in a clinical trial involving pediatric patients with TH, with or without associated cellular immunodeficiency. In addition to clinical outcomes and changes in thymic size, the study also assessed immunological parameters, including CD3+CD4+ and CD3+CD8+ T cell subsets, in patients with cellular immunodeficiency.
Materials and Methods
Study Design
This study was designed as a single-arm, phase III, multicenter, non-controlled, non-randomized clinical trial involving two groups of pediatric patients receiving treatment. The trial was prospectively registered in the Cuban Public Registry of Clinical Trials (ID code RPCEC00000247), a database recognized by the World Health Organization’s (WHO) International Clinical Trials Registry Platform.
The study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki, the Good Clinical Practice (GCP) guidelines, and the national regulatory standards established by the Center for State Control of Medicines, Equipment, and Medical Devices (CECMED, Cuba). The study protocol was approved by the Ethics Committee of the Institute of Hematology and Immunology (Cuba) on December 13, 2016. Written informed consent was obtained from the parents or legal guardians of all participants prior to enrollment.
Patient Selection
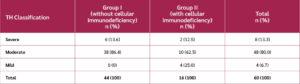
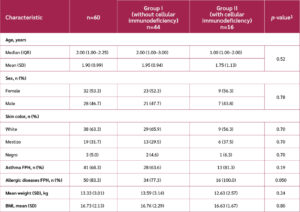
Children aged 1 to 5 years, of any gender, who attended the immunology service at the Institute of Hematology and Immunology and at the Pediatric Hospital “Angel Arturo Aballí”, were screened for inclusion. Patients were divided into two groups. Group I included children with TH and clinical manifestations of immunodeficiency, but without a confirmed laboratory diagnosis of cellular immunodeficiency. Group II included patients with TH, clinical symptoms, and a confirmed laboratory diagnosis of cellular immunodeficiency.
Thymic hypoplasia was diagnosed when the thymic area measured less than 1000 mm². Severity was classified as follows: severe for values less than 500 mm², moderate between 500 and 799 mm², and mild between 800 and 999 mm² (16). Cellular immunodeficiency was diagnosed based on age-specific reference values for CD3+CD4+ and CD3+CD8+ T cells, CD19+ B cells, CD3-CD56+ NK cells, and the CD4+/CD8+ T cell ratio, following the criteria of Comans-Bitter et al. (17).
Exclusion criteria included: a history of severe allergic reactions or severe generalized eczema; diagnosis of DiGeorge syndrome or other non-cellular immunodeficiencies (except IgA deficiency); steroid use within the previous 45 days; malignancy or autoimmune disease; chronic diseases such as type I diabetes mellitus (DM) or other decompensated endocrinopathy; severe congenital malformations requiring ongoing medical or surgical intervention; and hemoglobin levels below 9 g/L.
Treatment
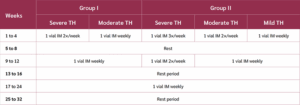
All patients received intramuscular (IM) Biomodulina T (3 mg; BIOCEN, Cuba). Treatment consisted of an initial 4-week cycle, followed by a 4-week rest period, and then a second 4-week cycle, again followed by a 4-week rest period. Within each treatment cycle, patients received 1, 2, or 3 weekly doses depending on TH severity (Table 1). Patients whose thymic size had not normalized by week 16 received a third treatment cycle of one IM vial per week for 8 weeks, followed by an additional 8-week rest period. Patients were evaluated at the end of weeks 8, 16, 24, and 32.
Concomitant treatments included oral vitamin C and vitamin A, administered daily throughout the study period. Allergic children were also given available oral antihistamines as needed.
Outcomes and Evaluations
The primary outcome of the study was the thymic area, assessed by ultrasonography using real-time mobile ultrasound devices (Philips Affiniti 70 and MINDRAY MCI) equipped with high-resolution pediatric transducers. Scans were performed in sagittal parasternal (angled from the upper edge of the second rib to the lower edge of the fourth rib at the sternum) and axial trans-sternal planes at the level of the sternal angle. All measurements were taken during the expiratory phase. The ultrasonography was performed at baseline, and at weeks 16 and 32. Secondary outcomes included the frequency and etiology of infections (categorized as bacterial, viral, fungal, or parasitic), antibiotic consumption, and hospital admissions.
A categorical composite variable termed the General Clinical Response Criterion (GCRC) was employed to assess overall treatment efficacy. Patients were classified as follows:
- Better: Normalized thymic area and ≥50% reduction in infection frequency compared to baseline
- Worse: Unchanged or reduced thymic area and stable or increased infection frequency
- Equal: Any outcome not meeting the criteria for “better” or “worse”
Lymphocyte subpopulations were quantified using flow cytometry (GALLIOS cytometer, Beckman Coulter, USA) with fluorochrome-conjugated monoclonal antibodies (anti-CD3-PC5, CD4-FITC, CD8-PE, CD56-APC; Dako, Denmark; Miltenyi Biotec, Germany). Serum levels of total IgG, IgM, and IgA were measured using immunoturbidimetric assays (SPAPLUS, The Binding Site, UK).
Statistical Analysis
The statistical hypothesis was based on the results of prior studies conducted in Cuba (13-15). Using a linear regression model, the expected slope of the thymic area was 45.9 ± 6.71 mm²/month. Sample size was calculated according to Dupont and Plummer (18) for regression-based designs, with α=0.05 and 1-β=0.9. Assuming a 60% increase in the thymic area at week 32, 55 patients were required. To account for potential dropouts, the final sample size was set at 60.
Statistical analyses were performed using SPSS version 25 (IBM Corp., Armonk, NY, USA) and GraphPad Prism version 8.0 (GraphPad Software, San Diego, CA, USA). Normality of data distribution was assessed using the Kolmogorov-Smirnov and Shapiro-Wilk tests to determine the suitability for parametric analysis. Continuous variables were analyzed using two-way ANOVA and mixed-effects models, as appropriate. Categorical variables were compared using Fisher’s exact test. A p-value <0.05 was considered statistically significant.
Results
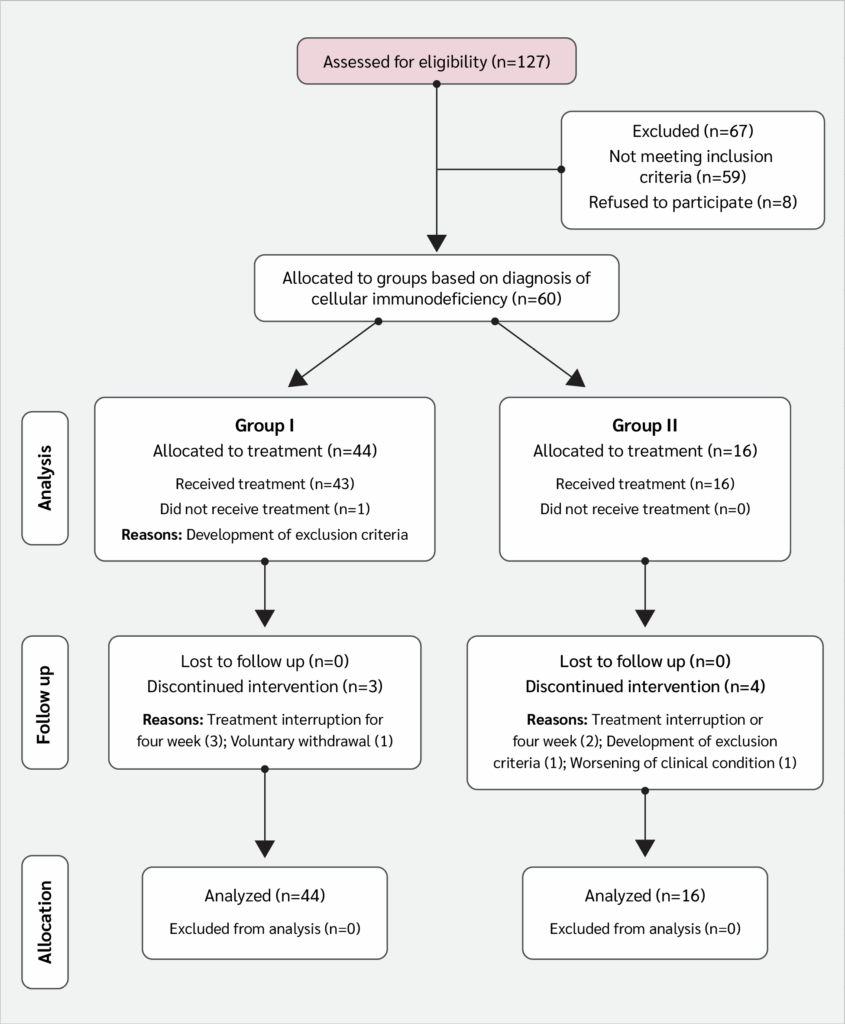
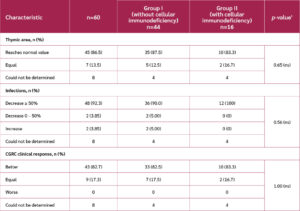
The study included 60 patients: 44 in Group I and 16 in Group II (Table 2). A total of 52 patients (86.6%) completed the treatment regimen and were evaluated at week 32, thus providing valid data on the primary variable (thymic area) (Figure 1, CONSORT flow diagram). There were eight treatment interruptions (13.3%), four in each group. The leading causes of interruption were voluntary withdrawal or worsening clinical conditions.
The median age of participants was 2 years. Baseline demographic characteristics and risk factors are shown in Table 3. Overall, no significant differences were observed between the groups, except for a higher frequency of personal history of allergic diseases in Group I. No significant differences in perinatal history were found between groups.
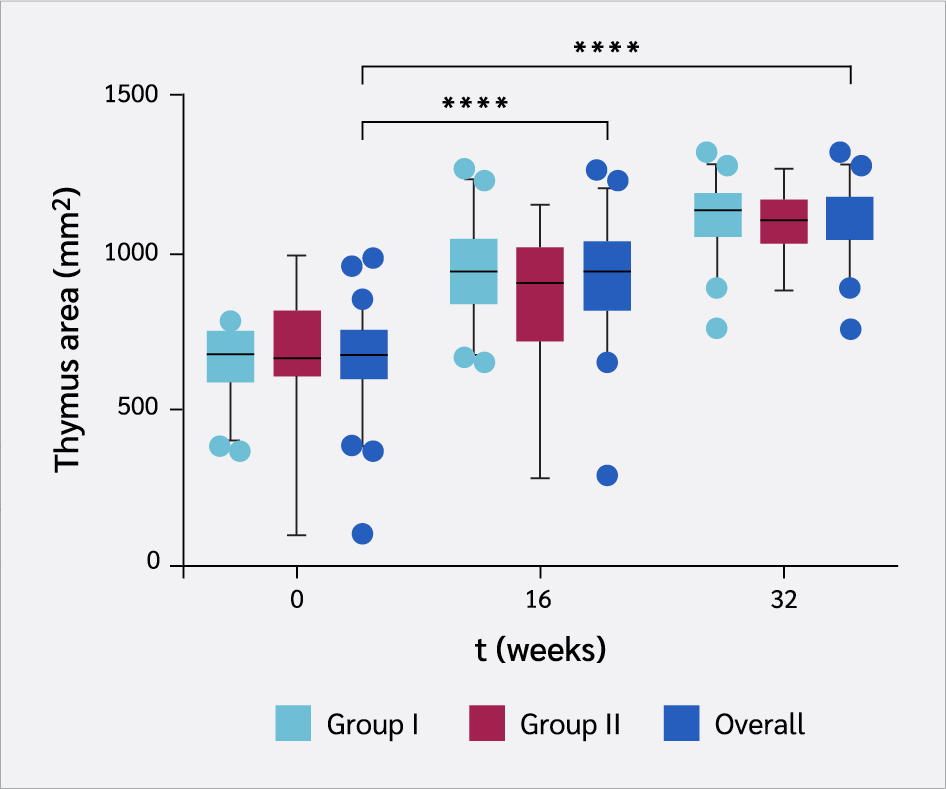
The thymic area increased as the treatment progressed, with a significant difference from baseline at week 16, and a more pronounced increase by week 32 (p<0.0001) (Figure 2). Both groups showed similar trends without significant intergroup differences. The mean increase by the end of treatment was 67% (95% CI 61–73%) compared to baseline. At the individual level, 45 of the 52 patients (87%) achieved a thymic area within the normal range, demonstrating high efficacy of the treatment for the primary outcome.
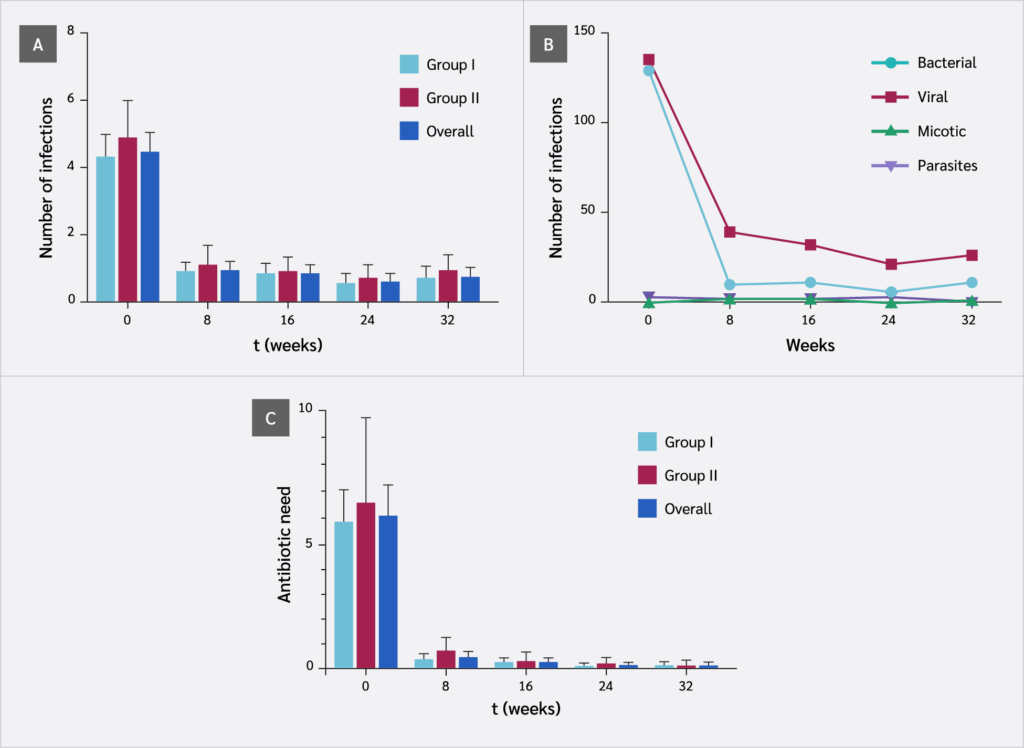
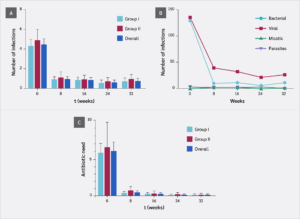
Consistent with the increase in thymic area, the frequency of infections decreased progressively, with a significant reduction observed as early as week 8 (p<0.0001), and no significant differences between groups (Figure 3A). The mean reduction in infection frequency was 84% (95% CI 70–95%) compared to the baseline. Bacterial and viral infections predominated; fungal and parasitic infections were rare. The reduction in bacterial and viral infections was significant by week 8 (p<0.0001), with a slightly greater effect observed for bacterial infections (Figure 3B). Correspondingly, antibiotic use declined significantly from week 8 onward (p<0.0001) (Figure 3C). While 96.5% of patients used antibiotics at baseline, this figure fell to 7.69% by week 32, with no hospital admissions reported.
The composite variable CGRC, which integrates thymic area and infection frequency, showed that no patients were classified as “worse”, and 82.7% were classified as “better”, with no significant differences between groups. According to the intention-to-treat analysis, which included patients who discontinued treatment, 71.7% demonstrated improvement based on the CGRC (Table 4).
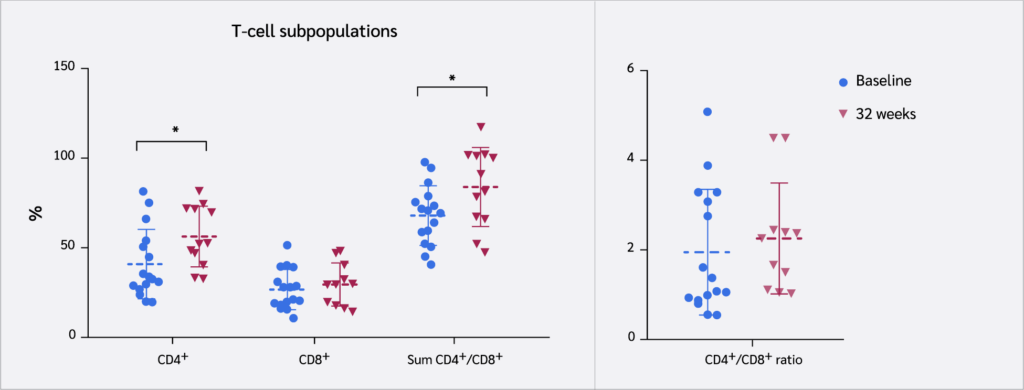
Evaluations of T cell markers (CD4+ and CD8+), as well as B lymphocytes (CD19+) and NK cells (CD56+), were performed in Group II at baseline and week 32. A significant increase was observed in the relative proportion of CD4+ T cells and the sum of CD4+ and CD8+ T cells (p) (Figure 4). The CD4/CD8 ratio showed an increasing trend, although it did not reach statistical significance. No significant changes were detected in B lymphocyte and NK cell populations (data not shown).
Among the enrolled patients, 13 had decreased serum IgA concentrations. A significant increase in IgA was observed, from a mean of 0.41 (±0.13) to 1.58 (±1.32) (p=0.013; Wilcoxon signed rank exact test) at week 32. Normal IgA values were restored in 4 patients (30.8%, p=0.012). No significant changes were observed in IgG and IgM levels.
Discussion
The present study evaluated the administration of the thymic polypeptide fraction Biomodulina T in children with TH. The results demonstrate not only clinical benefits, evidenced by a sharp reduction in infections associated with the increase and normalization of the thymic size, but also an increase in immunological markers such as the proportion of CD4+ T cells in peripheral blood and the concentration of serum IgA antibodies.
The thymus is highly active during the prenatal period and grows naturally after birth, primarily during the first 4 to 6 months of life (1,2). Although it continues to increase in weight until puberty, its proportional size relative to the mediastinum decreases. After puberty, the gland undergoes gradual physiological involution. The size of the thymus, as well as its growth, has been associated with prenatal nutrition, birth weight, and breastfeeding (1,2). Children with a small thymus in the first years of life often present clinically as immunodeficient. Currently, the measurement of the thymic area is a recognized tool in the clinical practice of pediatric immunology in Cuba (19-22). These findings support further exploration of the relationship between TH and infection susceptibility in children and align with the results observed in the present investigation.
The lack of a control group in this study could be a potential source of bias. However, a control group was not included due to ethical concerns; it was not considered appropriate to withhold a treatment already in routine clinical practice in Cuba, even if it is not yet an officially approved indication, particularly given the absence of alternative curative options. Nevertheless, the demonstrated relationship between clinical results and the increase in thymic size and with T cell-dependent immune markers in a subset of patients adds robustness to the conclusions. The observed rate of thymic enlargement was consistent with the statistical hypothesis of the study (an increase above 60% within 32 weeks), which was based on data from previous controlled studies conducted in Cuba between 1999 and 2007. Given the age range of the patients (1 and 5 years) and their previous clinical histories, the outcome could hardly be explained by a spontaneous growth of the gland, not associated with the intervention.
Thymic extracts contain several polypeptides capable of promoting T lymphocyte maturation (6). Among these, pro-thymosin α and its terminal fragment thymosin α1 have received special attention (22-25). Thymosin α1, also known as timalfasin, is an approved drug in several countries, indicated for immune reconstitution and the treatment of viral and bacterial infections, particularly in older adults. In addition to its immunopotentiating activity, pro-thymosin α plays a role in intracellular processes related to cell growth (23). Another major component of thymic extracts, thymosin β4, has a powerful tissue regenerative action (26). The exact mechanisms of thymic involution and its reversal remain poorly understood. Experimental data from mouse models suggest that thymic stromal cells, rather than hematopoietic cells, drive thymic involution (27,28). Other substances, such as growth hormone, have also shown efficacy in immune system reconstitution and enhancement of thymic mass in HIV patients (28). These pharmacological activities of thymic polypeptides seem to be consistent with the findings of the present study, supporting both immune responses and thymic growth.
Previous evaluations of Biomodulina T have investigated its immunological mechanisms of action, primarily in older adults and oncology patients (9-12). However, previous clinical research in children did not address its immunological effects. In older adults, Biomodulina T has been shown to significantly increase naïve T cells (both CD4+ and CD8+), as well as CD19+ B lymphocytes and CD56+ NK cells, the latter being key components of innate antiviral immunity. In a recent clinical trial conducted in nursing homes in Cuba, Biomodulina T reduced COVID-19 lethality in a preventive setting (11). In the present study, results from children, particularly those with cellular immunodeficiency, demonstrated an increase in CD4+ T cells and a non-significant trend toward increased CD8+ T cells. The CD4/CD8 ratio also showed a non-significant tendency to increase within the normal range. The small sample size (only 12 patients) and variability of flow cytometry measurements limited statistical precision. Nevertheless, these results are consistent with previous findings in older adults. An increased CD4/CD8 ratio has been associated with improved responses to bacterial and viral infections (29). The results also demonstrated clinical efficacy against viral infections, although no significant effect was observed on CD3-CD56+ NK cells.
Of particular relevance is the observed increase in IgA antibodies, which are an important component of the adaptive immune response at the mucosal level. The association between decreased IgA levels and reduced thymic size may be explained by the immaturity of the immune system during the first year of life and the role of Th cells in antibody class switching (30). In addition, the high prevalence of allergic conditions in this study, particularly in the group with cellular immunodeficiency (with more than half of Group I and all of Group II exhibiting allergic symptoms), constitutes a predisposing factor for reduced IgA levels.
Few studies have reported the use of thymic extracts or synthetic thymic peptides in the treatment of aplasia or TH in children. An early study in 1989 provided preliminary evidence of clinical benefit in a small group of children with DiGeorge syndrome (31). De Mattia et al. (32) administered a bovine thymic extract to pediatric patients with recurrent infections and observed a statistically significant improvement in lymphocyte-dependent antibacterial activity, along with clinical benefits in 3-year-old children.
In Cuba, several pioneering studies have investigated children with TH, including populations similar in age to those in the present trial (13-15). In a 1996 study involving children aged 8 months to 6 years with recurrent infections, the effect of Biomodulina T (34 patients) was compared to that of Levamisole. Biomodulina T induced a mean increase in thymic area from 520.38 mm² to 907.35 mm², representing a 74.3% increase (14). In a subsequent report, eutrophic children with recurrent infections and a thymic area <1000 mm² were studied. Patients treated with Biomodulina T consistently showed a greater increase in thymic area, from 603.02 mm² to 1003.62 mm², a 66% increase (15). In the present study, 92.3% of patients showed clinical improvement based on reduced infections, and 82.7% showed improvement according to the CGRC variable, which also accounted for thymic size. The mean increase in thymic area was 67%, closely aligning with previously reported values. These findings confirm the clinical efficacy of Biomodulina T in the TH treatment and provide new evidence of improvements in adaptive immunity markers that support the observed clinical outcomes.
An important contribution of the current research lies in its methodological framework as a phase III clinical trial, with prospective protocol approval and compliance with GCP guidelines throughout its execution. Accordingly, the results are valid not only in terms of their contribution to scientific knowledge but also for their potential application in clinical practice in Cuba, supporting the official approval of a new medical indication (treatment of TH in children), based on evidence. Although the sample size was limited due to the low prevalence of the condition, the results provide confidence in both clinical and immunological parameters. Future observational studies are needed to expand and refine these findings, particularly in terms of safety.
Conclusion
This clinical trial confirms the efficacy of Biomodulina T in treating TH in children aged 1 to 5 years. The treatment resulted in normalization of thymic size, suppression of bacterial and viral infections, and enhancement of adaptive cellular immunity, including Th cells and IgA antibodies in immunodeficient patients. The observed clinical benefits are consistent with the immunological changes and the known pharmacological effects of the thymic polypeptides contained in the product.
Ethical Approval
The Ethics Committee of the Institute of Hematology and Immunology, Havana, Cuba approved the study protocol on December 13, 2016 with approval number 2016.12.13/01.
Informed Consent
Written informed consent was obtained from parents or legal guardians of all participants prior enrollment.
Peer-review
Externally peer-reviewed
Author Contributions
Concept – O.M.G.P., A.L.R., M.C.R.Z., C.M.A., V.M.S.; Design – O.M.G.P., M.C.R.Z., A.L.R., C.M.A., K.R.G.; Supervision – C.M.A., A.L.R.; Fundings – M.C.R.Z., A.L.R.; Materials – M.C.R.Z., C.M.A., K.R.G., L.R.V., O.M.G.P.; Data Collection and/or Processing – O.M.G.P., K.R.G., V.M.S., L.R.V.; Analysis and/or Interpretation – O.M.G.P., A.L.R., L.R.V., V.M.S.; Literature Review – O.M.G.P., A.L.R.; Writer – O.M.G.P., A.L.R.; Critical Reviews – C.M.A., M.C.R.Z., V.M.S., K.R.G.
Conflict of Interest
The authors declare no conflict of interest.
Financial Disclosure
The authors declared that this study has received no financial support.
References
Shichkin VP, Antica M. Key factors for thymic function and development. Front Immunol. 2022;13:926516. [CrossRef]
Duah M, Li L, Shen J, Lan Q, Pan B, Xu K. Thymus degeneration and regeneration. Front Immunol. 2021;12:706244. [CrossRef]
Sastrowijoto SH, Vandenberghe K, Moerman P, Lauweryns JM, Fryns JP. Prenatal ultrasound diagnosis of rhizomelic chondrodysplasia punctata in a primigravida. Prenat Diagn. 1994;14(8):770-6. [CrossRef]
Akar H, Konuralp C, Baysal K, Kolbakir F. Ellis-van Creveld syndrome associated with thymic hypoplasia. Asian Cardiovasc Thorac Ann. 2002;10(4):336-8. [CrossRef]
Pérez M, Fundora H, Notario M, Rabassa J, Hernández MA, Rodríguez A. [Immunoepidemiological risk factors in children with recurrent respiratory infections]. Rev Cubana Pediatr. 2011;83(3):225-35. Spanish.
Low TL, Goldstein AL. Thymosins: structure, function and therapeutic applications. Thymus. 1984;6(1-2):27-42.
Rodriguez Martin RR, Gonzalez Gonzalez O, Rodriguez Gonzalez C, Rodriguez Gonzalez RR. History of the discovery and development of Biomodulina T (InmunyVital®), a useful immunomodulator with a broad range of clinical applications. Int Immunopharmacol. 2023;119:110167. [CrossRef]
Zamora MCR, Chávez SR, Almarales RL, Iglesias IR, Orta IA, López GJ, et al. [Safety of Biomodulina T® in the Cuban population before and during its use in the prevention of COVID-19]. Rev Habanera Cienc Med. 2023;22(2):7. Spanish.
Saavedra D, Fuertes SA, Suárez GM, González A, Lorenzo-Luaces P, García B, et al. Biomodulina T partially restores immunosenescent CD4 and CD8 T cell compartments in the elderly. Exp Gerontol. 2019;124:110633. [CrossRef]
Ramos EH, Suárez VM, Hernández IC, Gomez RP, Rivera DG, Zamora MCR, et al. Effect of Biomodulina-T® and VA-MENGOC-BC® on lymphocyte subpopulations in older adults. Exp Gerontol. 2021;153:111497. [CrossRef]
Hernández IC, Suárez VM, Ramos EH, Marrero YT, Domínguez GD, Pérez YD, et al. BIOMODULINA T® modulates lymphocyte compartments in institutionalized Cuban geriatric patients. J Cellular Immunology. 2022;4(2):79-91. [CrossRef]
Saavedra D, Añé-Kourí AL, Ledón N, Crombet T, Lage A. Treatment with the thymic polypeptide fraction Biomodulina T potentiates immune responses in older adults and cancer patients: an overview of the most recent studies. Ageing Cancer Res Treat. 2024;1:80. [CrossRef]
Mollineda O, Martínez A, Rabaza J, Hernández CM, Romero JM. [Immunomodulators. Respose of under one-year-old malnourished infants to immunomodulatory therapy]. Rev Cubana Pediatr.1999;71(4):191-6. Spanish.
Christian López L de la C, Rodríguez Marín RR, Rabassa Pérez J, Santamaría Lafargue M, Romero del Sol JM, González Ross E. [Effect of Biomodulina T 1000 on the thymus in children with recurrent infections]. Rev Cubana Pediatr. 2000;72(1):3-9. Spanish.
Christian López LC, Rabassa Pérez J, Tamayo V, Marzoa Silva N, Rodríguez Álvarez I, Sosin Curbelo AM. [Therapeutic effect of homeopathic Biomodulina in children with recurrent infections]. Gaceta Homeopática de Caracas. 2006;14(2):12-22. Spanish.
Rabassa Pérez J, Christian López LC, Martínez Morales A, Romero del Sol JM, Sosa L. [Sonographic evaluation of the thymus in healthy children: a preliminary study]. Rev Cubana Pediatr. 2004;76(3):1-7. Spanish.
Comans-Bitter WM, de Groot R, van den Beemd R, Neijens HJ, Hop WC, Groeneveld K, et al. Immunophenotyping of blood lymphocytes in childhood. Reference values for lymphocyte subpopulations. J Pediatr. 1997;130(3):388-93. [CrossRef]
Dupont WD, Plummer WD Jr. Power and sample size calculations for studies involving linear regression. Control Clin Trials. 1998;19(6):589-601. [CrossRef]
Jeppesen DL. The size of the thymus: an important immunological diagnostic tool? Acta Paediatr. 2003;92(9):994-6.
de la Guardia Peña OM. [Thymic hypoplasia in an infant with cystic fibrosis: a case report] Rev Cuba Hematol Inmunol Hemoter. 2014;30(1):74-80. Spanish.
Rabassa Pérez J, Fundora H, Rodríguez A, Hernández MA. [Ultrasonography of spleen and thymus as an immuneresponse evaluator in children with recurrent infections] VacciMonitor. 2010;19(2):5-10. Spanish.
de la Guardia Peña OM. [Algorithm for pediatric thymic hypoplasia diagnosis and treatment, associated or not with immunodeficiency] Rev Cuba Hematol Inmunol Hemoter. 2022;38(1):1-2. Spanish.
Hannappel E, Huff T. The thymosins. Prothymosin alpha, parathymosin, and beta-thymosins: structure and function. Vitam Horm. 2003;66:257-96. [CrossRef]
Samara P, Ioannou K, Tsitsilonis OE. Prothymosin alpha and immune responses: Are we close to potential clinical applications? Vitam Horm. 2016;102:179-207. [CrossRef]
Dominari A, Hathaway Iii D, Pandav K, Matos W, Biswas S, Reddy G, et al. Thymosin alpha 1: A comprehensive review of the literature. World J Virol. 2020;9(5):67-78. [CrossRef]
Bock-Marquette I, Maar K, Maar S, Lippai B, Faskerti G, Gallyas F Jr, et al. Thymosin beta-4 denotes new directions towards developing prosperous anti-aging regenerative therapies. Int Immunopharmacol. 2023;116:109741. [CrossRef]
Silva CS, Reis RL, Martins A, Neves NM. Recapitulation of thymic function by tissue engineering strategies. Adv Healthc Mater. 2021;10(20):e2100773. [CrossRef]
Cowan JE, Takahama Y, Bhandoola A, Ohigashi I. Postnatal involution and counter-involution of the thymus. Front Immunol. 2020;11:897. [CrossRef]
Bruno G, Saracino A, Monno L, Angarano G. The Revival of an "old" marker: CD4/CD8 Ratio. AIDS Rev. 2017;19(2):81-8.
de la Guardia Peña OM, Abraham CM, Ruiz Villegas LR. [Association between thymic hypoplasia and decreased immunoglobulin A]. Rev Cuba Hematol Inmunol Hemoter. 2021;37(1):1-10. Spanish.
Barrett DJ, Wara DW, Ammann AJ, Cowan MJ. Thymosin therapy in the DiGeorge syndrome. J Pediatr. 1980;97(1):66-71. [CrossRef]
De Mattia D, Decandia P, Ferrante P, Pace D, Martire B, Ciccarelli M, et al. Effectiveness of thymostimulin and study of lymphocyte-dependent antibacterial activity in children with recurrent respiratory infections. Immunopharmacol Immunotoxicol. 1993;15(4):447-59. [CrossRef]
VOLUME
,
ISSUE
Correspondence
Received
Accepted
Published
Suggested Citation
DOI
License