Relationship Between CD73 Expression on T Cells and Response to Treatment in De-novo Adult Acute Myeloid Leukemia Patients in Egypt
Abstract
Objective:
Acute myeloid leukemia (AML) is a heterogeneous blood malignancy with wide genomic aberrations, treatment approaches, and treatment outcomes. Successful treatment for AML remains challenging. CD73 is a recently recognized immune checkpoint mediator that is highly expressed in tumor microenvironment. It catalyzes the hydrolysis of adenosine monophosphate (AMP) to adenosine, which subsequently inhibits anti-tumor immune responses. This study aimed to investigate the relationship between CD73 and the response to induction therapy in adult Egyptian patients with AML.
Materials and Methods:
A total of 44 blood samples were collected from 25 patients. Each survivor of induction therapy provided two samples: one on the day of initial diagnosis and another on the day of assessment of response to induction therapy (ARIT). Patients who did not survive induction therapy provided only one sample on the day of diagnosis. CD73 was measured on T cells using flow cytometric analysis and correlated with the response to treatment.
Results:
Among the study cohort, median CD73 expression was 10.6% on T cells and 22.1% on CD8+ T cells. There was no statistically significant difference across different risk categories, nor was there an association with treatment response. However, CD73 expression showed a positive correlation with 28-day survival.
Conclusion:
CD73 expression did not show a significant relationship with any patient characteristics or disease parameters and did not affect treatment response. Nevertheless, it was positively correlated with 28-day survival.
Keywords:
De-novo, acute, myeloid, leukemia treatment, outcome CD73Introduction
Acute myeloid leukemia (AML) is the most common form of acute leukemia in adults, with an estimated 20,380 new cases and 11,310 deaths reported in the USA in 2023 alone (1). According to the National Cancer Institute, only 31.7% survive beyond five years post-therapy (2). Treatment of patients with AML consists of three phases: induction, consolidation, and maintenance. The induction phase, as outlined by the National Comprehensive Cancer Network (NCCN), typically involves the u00223+7u0022 protocol, which consists of cytarabine (200 mg/m²/day) administered as a continuous intravenous infusion for seven days, combined with doxorubicin (25 mg/m²/day) given intravenously for three consecutive days (3). Following induction therapy, a follow-up bone marrow biopsy is performed to confirm whether remission has been achieved before proceeding to consolidation therapy (4). Recently, improvements have been made to the induction chemotherapy regimen, resulting in higher complete remission (CR) rates and better relapse-free and overall survival. However, focusing research efforts to achieve better long-term outcomes is still required (5).
Immune checkpoint molecules are highly expressed in the tumor microenvironment (TME), where they can modulate the response of T cells to tumor antigens and influence the patient's clinical outcome (6). CD73 is one of the recently recognized immune checkpoint receptors that is expressed on various immune and non-immune cells in the TME, including T cells, B cells, natural killer cells, dendritic cells, myeloid-derived suppressor cells, and tumor-associated macrophages. It exhibits 5′-nucleotidase activity and can hydrolyze adenosine monophosphate (AMP) to adenosine, which has immunosuppressive effects. Consequently, CD73 plays an essential role in balancing inflammation and immune suppression. Additionally, it can modulate tumorigenesis, proliferation, migration, and immune escape; therefore, it can be examined as part of tumor immunotherapy (7).
Recently, immune checkpoint therapy has demonstrated remarkable clinical outcomes for many solid tumors. It has been tested in the treatment of blood malignancies such as Hodgkin’s lymphoma and has shown promising improvements in treatment outcomes (8). Currently, antibody therapy targeting CD73 is being tested in many clinical trials, either alone or in combination with other small molecular antagonists (ClinicalTrials.gov Identifiers: NCT04797468, NCT04148937, NCT03616886) (9).
The crucial role of CD73 in the therapeutic landscape of AML needs further validation. The current study aimed to investigate the relationship between CD73 expression on T cells and the treatment outcome of induction therapy in adult Egyptian patients with AML.
Materials and Methods
This prospective observational study was conducted on 25 newly diagnosed AML patients who were admitted to the Clinical Hematology and Oncology Unit, Department of Internal Medicine, University Hospital, Cairo, Egypt, and the Nasser Institute Hospital for Research and Treatment, Cairo, Egypt. The study period spanned from October 2021 to October 2023.
Patient Selection
Inclusion criteria:
- Newly diagnosed (de-novo) AML patients
- Age between 18 and 65 years
- Patients deemed fit for intensive chemotherapy of curative intent (i.e., free from cardiac, renal, or liver impairment)
Exclusion criteria:
- Age <18 or >65 years
- Relapsed AML patients
- Patients with AML who had chemotherapy initiated before enrollment in the study
- Secondary AML with a preceding hematologic disorder
- Diagnosis of acute promyelocytic leukemia (APL)
- Any acute leukemia other than AML
- History of solid malignancies
- Patients who were considered ineligible to receive intensified treatment
Clinical Evaluation and Sample Collection
A comprehensive history was taken from the patients, including demographic data, clinical data, routine laboratory tests, such as complete blood count (CBC) and C-reactive protein (CRP), and an assessment of comorbidities were done.
For flow cytometric analysis, 3 mL of peripheral blood was withdrawn from each participant under complete aseptic conditions in ethylene-diamine-tetra-acetic acid (EDTA) tube. Blood samples were collected from each patient twice, at the time of diagnosis and on the day of assessment of response to induction therapy (ARIT). The tubes were transferred immediately to Ain Shams University Hospital laboratory for flow cytometric analysis.
Ethical Approval
The study protocol was approved by the Ethical Committee for Scientific Research, Faculty of Medicine, Ain Shams University (FWA 000017585 / MD 379 / 2019). Written informed consent was obtained from all patients or their legal guardians after providing detailed information about the study objectives and procedures.
Flow Cytometric Analysis
CD45, CD3, CD4, CD8, and CD73 monoclonal antibodies (Beckman Coulter, USA) were used to detect
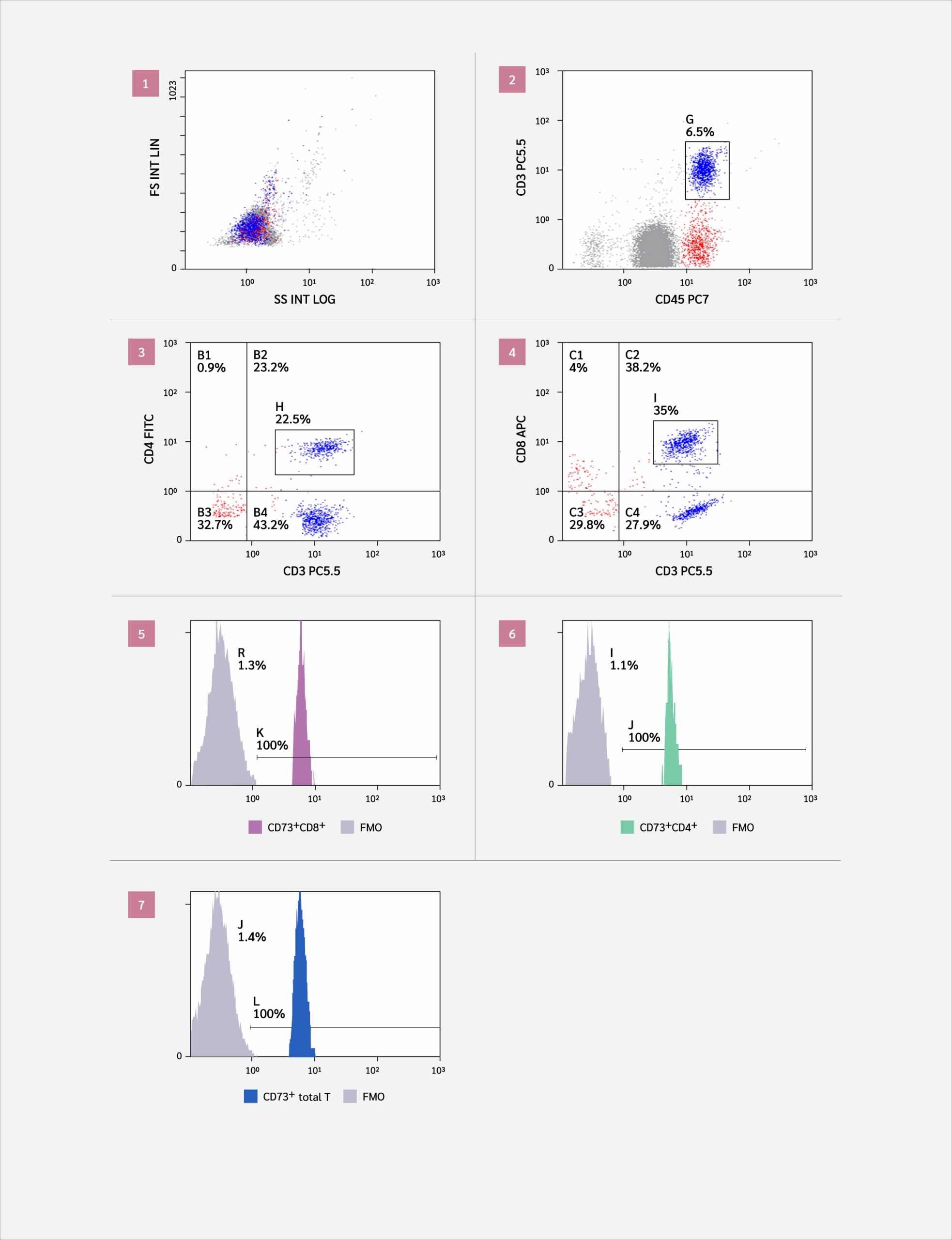
CD73+CD4+ T cells and CD73+CD8+ T cells. Overall, 50 μL of EDTA-treated blood was added to each of the sample tubes, and then 5 μL of each of the following anti-human monoclonal antibodies was added: anti-CD45-PC7, anti-CD3-PC5.5, anti-CD4-FITC, anti-CD8-APC, and anti-CD73-PE. After incubation for 15 minutes at room temperature, protected from light, 1–2 mL of ammonium chloride-based erythrocyte lysing solution was added to each tube. Tubes were vortexed and then analyzed using the NAVIOS flow cytometer and the NAVIOS CXP software (Beckman Coulter, USA). The gating strategy is illustrated in Figure 1. A total of 3894 cells were analyzed in 182.3 seconds.
Data Management and Analysis
Statistical analysis was performed using SPSS 23.0 statistical software (IBM Corp., Armonk, NY, USA). Non-parametric quantitative data were presented using the interquartile range (IQR) and median. The chi-square, Mann-Whitney, Kruskal-Wallis test, and Wilcoxon signed-rank test were used to compare the differences between the two groups. The correlation of paired data was analyzed using Spearman’s correlation coefficient. A p-value of <0.05 was considered statistically significant.
Results
Demographic data
The study cohort had a median age of 41 years (range: 21–60) and consisted of 14 males (56%) and 11 females (44%). Eighty percent of the patients had no comorbidities, while 20% (n=5) had comorbid diabetes mellitus and hypertension.
Clinical information
According to the French-American-British (FAB) classification, the distribution of AML subtypes was as follows: M0 (n=4), M1 (n=6), M2 (n=7), M4 (n=5), and M5 (n=3). Cytogenetic studies were performed according to the European Leukemia Net (ELN) recommendations for the diagnosis and management of AML (5). Based on ELN, 5 (20%) patients were classified as being at favorable risk, 13 (52%) at intermediate risk, and 7 (28%) at adverse risk. Almost all patients (96%) had no extramedullary infiltration, while only one patient had an extramedullary infiltration in the lung, presenting hemorrhagic pleural effusion.
All the patients received induction chemotherapy. However, 6 (24%) patients died before completing the induction phase: five due to septic shock and one from neutropenic enterocolitis. The patients were categorized into 15 (78.9%) responders and 4 (21.1%) with refractory disease. Patients were followed for 28 days starting from the initiation of induction therapy. During this period, one additional patient died of septicemia. By the end of the follow-up period, 18 patients (72%) remained alive (Table 1).
Immunophenotyping
Table 2 shows that the median CD73 expression on total T cells was 13.4% (range: 0–100%). CD8+ T cells exhibited a higher median expression level of CD73 at 22.1%, compared to CD4+ T cells, which had a median expression of only 5.6%.
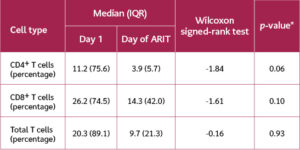
Table 3 presents a comparison of CD73 expression levels on total T cells, CD4+ T cells, and CD8+ T cells among the 19 patients who survived induction therapy. Expression was measured on the day of initial diagnosis and again on the day of ARIT. It was evident that the median level of CD73 expression decreased across all T cell subsets following treatment. However, this decrease was not statistically significant, with p-values of 0.93 for total T cells, 0.06 for CD4+ T cells, and 0.10 for CD8+ T cells.
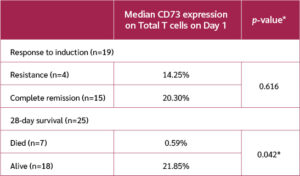
Among patients who survived the induction chemotherapy regimen (n=19), 15 responded to chemotherapy, achieving remission, while four were refractory. A comparison of responders versus non-responders in terms of CD73 expression revealed that the responder group had a higher median CD73 expression (20.3%) than non-responders (14.25%) (Table 4). However, the difference did not reach statistical significance (p=0.616).
A comparison between survivors (n=18) and deceased patients (n=7) based on their CD73 expression on the day of initial diagnosis revealed a significant difference. Survivors exhibited a markedly higher median CD73 level (21.85%) compared to deceased patients (0.59%), with a statistically significant p-value of 0.042 (Figure 2).
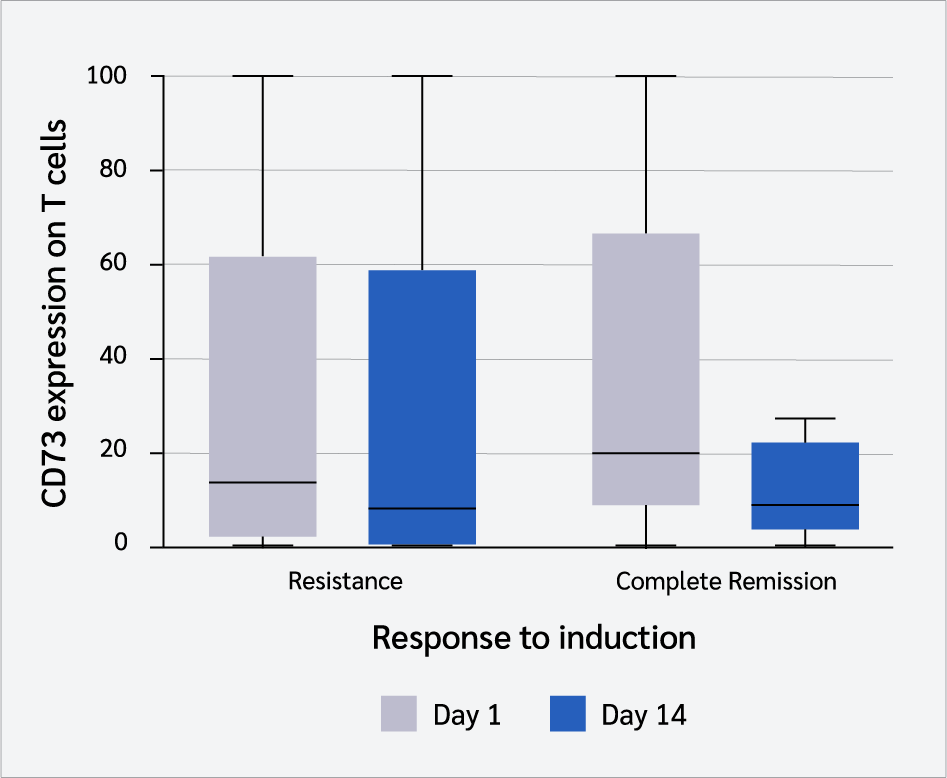
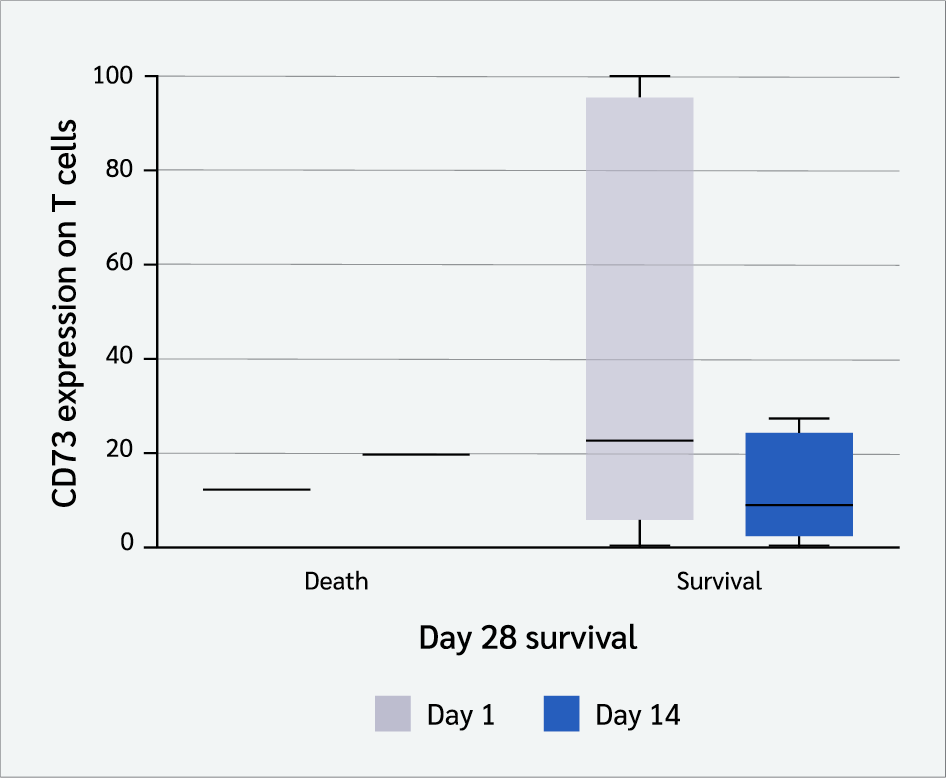
Figure 3 shows a comparison between CD73 expression level on total T cells on Day 1 and Day 14 after induction start in 28-day survivors and non-survivors.
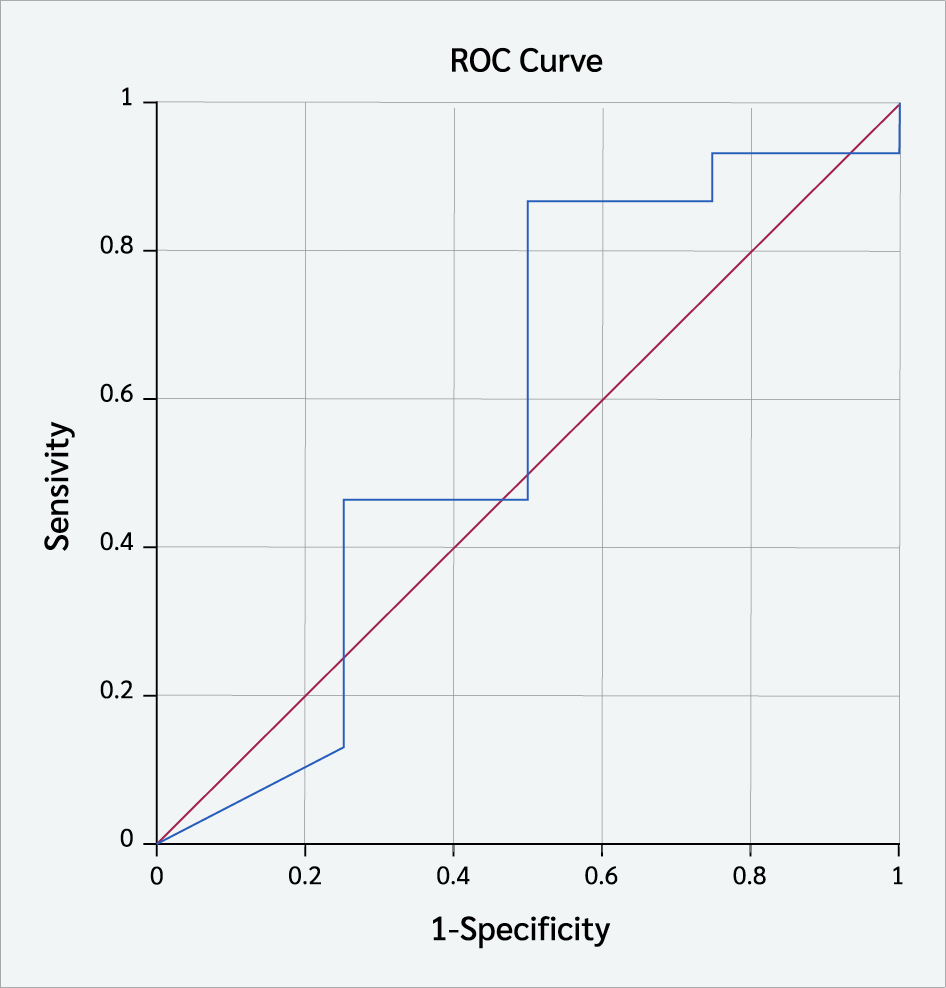
The optimal cut-off value of CD73 expression on total T cells at diagnosis for predicting complete remission was >5.650 (Figure 4). The area under the ROC curve (AUC) for CD73 was 0.583. The sensitivity and specificity were 86% and 50%, respectively.
When comparing the incidence rate of high CD73 expression on total T cells (defined as >median expression levels) between the responder and non-responder groups, the responder group exhibited a higher rate (approximately 60%). Similarly, the incidence of high CD73 expression on total T cells was also higher in the survivor group compared to the deceased group, at 61.1% and 14.3%, respectively.
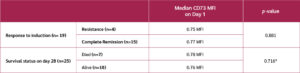
Median expression level of CD73 mean fluorescence intensity on total T cells on the day of initial diagnosis washigher in the responder group than in non-responders (p=0.881; Table 6).
Discussion
Although CD73 is expressed on many cells in the TME, the effector T cell is the primary cell responsible for the direct recognition and killing of tumor cells. Several studies assessed the functions of T cells in AML by high-dimensional immunophenotyping, gene expression, and functional studies (10,11) to characterize T cell function at the time of initial diagnosis and after induction chemotherapy. They found that multiple aspects of T cell function were deranged at diagnosis, with exhaustion and senescence being the dominant processes that reverted upon attaining complete remission.
The primary aim of this prospective study was to investigate the relation between CD73 expression on T cells and the treatment response to induction therapy in adult Egyptian patients with AML.
Regarding CD73 expression, our findings were consistent with a study conducted on 27 newly diagnosed AML patients in the USA, which reported a CD73 expression level of 22.26% on CD8+ T cells (12). However, a study from Germany reported a higher CD73 expression level on CD8+ T cells (41.35%) (13).
In studies assessing CD73 expression in solid tumors, methods other than flow cytometry have been predominantly used, such as immunohistochemistry and immunofluorescence. Notably, the criteria for defining high CD73 expression varied across IHC-based studies. For instance, some studies defined high CD73 expression as the percentage of positive-staining tumor cells exceeding the median expression level, while others considered staining intensity greater than 10% of positive-staining tumor cells as indicative of high CD73 expression (14). To align with these approaches, our research used the median expression level of CD73 on total Tcellsbefore the start of induction therapy (10.6%) as the cut-off value for determining high CD73 expression. This cut-off was specifically chosen to match the approach taken in previous studies, all of which examined CD73 expression in tumors before treatment. Patients whose CD73 expression met or exceeded this cut-off were classified as having high CD73 expression.
A study conducted in China on classical Hodgkin lymphoma patients reported convergent results, finding that CD73 was highly expressed in 40% of cases, as determined by immunohistochemical staining of CD73 in lymphoma samples (15). However, a higher incidence rate of high CD73 expression (61.7%) was reported by Ren et al. (16), who conducted immunohistochemical staining of CD73 in human head and neck squamous cell carcinoma (HNSCC) tissues from 162 patients with HNSCC in China. In contrast, other studies on bladder cancer and renal cell carcinoma reported lower incidence rates of high CD73 expression, at 26.4% and 15.9%, respectively, using immunohistochemical staining of CD73 in primary urothelial carcinoma tissue samples and nephrectomy tissue samples respectively (17,18). These discrepancies in CD73 expression levels can be attributed to differences in tumor types, assessment methods, and the criteria used for defining high CD73 expression across the studies.
Upon assessing the relationship between CD73 expression and various demographic data, our results were in concordance with those of a meta-analysis that included 14 studies with a total of 2951 patients representing 12 different types of cancers. In their study, they noted no correlation between high CD73 expression and age or sex (14).
Concerning the comparison of CD73 expression in different AML immunophenotypes, our result was contrary to the study conducted by Zhang et al. (19), which reported a positive association between CD73 expression and FAB. The difference between this study and theirs may be attributed to the different types of samples analyzed, as they assessed CD73 expression on blast cells rather than on peripheral blood mononuclear cells, as was done in our study. Additionally, the larger sample size utilized in their study may also contributed to the observed differences.
Upon comparing the level of CD73 expression on total T cells, CD4+ cells, and CD8+ cells at diagnosis and during the assessment of response to treatment, our results were in concordance with those of a study conducted in Brazil on 33 patients with breast cancer (20). They observed a significant reduction of CD73 activity between the samples at the time of diagnosis and 6 months after treatment. However, contradictory results were reported by another experimental study on mice (21), which found that CD73 expression increased after radiotherapy compared to the baseline. These discrepancies in results may be attributed to the nature of the research, which was experimental versus prospective human study, the type of malignancy studied, as well as the different methodologies used for measuring CD73 expression, which might explain, in part, the different outcomes.
The present study demonstrated a statistically significant positive association between CD73 expression and 28-day overall survival. Similarly, positive CD73 expression was strongly correlated with longer overall survival (OS) in studies conducted on bladder cancer and breast cancer (17,22). However, Jiang et al. (14) concluded in their meta-analysis that high CD73 expression was significantly correlated with shorter OS in various cancers. Also, a more recent study conducted on 215 patients with colorectal cancer liver metastasis in France noticed that patients with high levels of serum CD73 had shorter disease-specific survival (23).
Regarding the comparison between CD73 expression and the response to treatment, our results were in concordance with those noted in a study conducted on classical Hodgkin lymphoma patients, which found that the increased expression of CD73 was associated with a better response to treatment (15). On the other hand, a study reported opposite results, finding that increased expression of CD73 was associated with a lower response to treatment in head and neck cancer patients (24).
Despite the well-known immune-suppressive role of CD73 expression on tumor and stromal cells within the tumor microenvironment, the literature focusing on CD73 expression on T cells and its relationship with prognosis is limited. The positive prognostic impact of high CD73 expression in our study is supported by a striking observation made by Kong et al. (12), who found that CD73 expression on CD8+ T cells was associated with an enhanced immune response. CD73-CD8+ T cells exhibited greater functionality, while CD73-CD8+ T cells showed features of exhaustion, such as high expression of inhibitory receptors like PD-1 and TIGIT, reduced cytokine production capacity, and increased susceptibility to apoptosis. Additionally, our results may differ due to the type of tumor investigated, whereas our study focused on blood malignancies, while the aforementioned studies primarily examined solid tumors. Moreover, while those studies mainly measured CD73 expression on tumor cells, stromal cells, and Tregs, our study predominantly assessed CD73 expression on T cells. Therefore, understanding the specific distribution pattern of CD73 in each cancer type or disease status is crucial for the optimal design of clinical studies targeting CD73 in cancer treatment.
This study has several limitations, including the small number of patients enrolled, the shorter follow-up period, and the absence of a control group. As such, further studies on a larger scale of AML patients are recommended to assess the expression of CD73 on T lymphocytes. Additionally, longer follow-up studies with serial assessment of CD73 levels will be necessary to determine its utility as a predictive indicator for treatment response in AML patients.
Conclusion
Although no statistically significant association was found between CD73 expression on T cells and disease parameters or treatment response, it was positively correlated with 28-day survival, suggesting that CD73 may serve as a prognostic marker in AML. Research is required to confirm this hypothesis.
Ethical Approval
The study protocol was approved by the Ethical Committee for Scientific Research, School of Medicine, Ain Shams University (FWA 000017585 / MD 379 / 2019).
Peer Review
Externally peer-reviewed
Author Contributions
Concept – N.M.S.A., S.E.M.T., H.M.A.H., G.M.F.; Design – E.I.A.M., S.E.M.T., H.M.A.H., G.M.F.; Supervision – F.E.Y.F.; Fundings – E.I.A.M.; Materials – E.I.A.M.; Data Collection and/or Processing – E.I.A.M.; Literature Review – N.M.S.A., S.E.M.T., F.E.Y.F.; Writer – E.I.A.M., N.M.S.A., F.E.Y.F. Critical Reviews – H.M.A.H., G.M.F.
Conflict of Interest
The authors declare no conflict of interest.
Financial Disclosure
The authors declared that this study has received no financial support.
References
Wysota M, Konopleva M, Mitchell S. Novel therapeutic targets in acute myeloid leukemia (AML). Curr Oncol Rep. 2024;26(4):409-20. [CrossRef]
National Cancer Institute (U.S.). Acute myeloid leukemia treatment (PDQ®)–health professional version. Incidence and mortality [Internet]. Bethesda (MD): National Cancer Institute; 2024 [cited November 3, 2024]. Available from: https://www.cancer.gov/types/leukemia/hp/adult-aml-treatment-pdq
Tallman MS, Wang ES, Altman JK, Appelbaum FR, Bhatt VR, Bixby D, et al. Acute Myeloid Leukemia, Version 3.2019, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2019;17(6):721-49. [CrossRef]
Yildizhan H, Barkan NP, Turan SK, Demiralp Ö, Demiralp FDÖ, Uslu B, et al. Treatment strategies in cancer from past to present. In: Grumezescu AM, editor. Drug targeting and stimuli sensitive drug delivery systems. Cambridge: Elsevier; 2018. p. 1-37.
Döhner H, Wei AH, Appelbaum FR, Craddock C, DiNardo CD, Dombret H, et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood. 2022;140(12):1345-77. [CrossRef]
Tang K, Zhang J, Cao H, Xiao G, Wang Z, Zhang X, et al. Identification of CD73 as a novel biomarker encompassing the tumor microenvironment, prognosis, and therapeutic responses in various cancers. Cancers (Basel). 2022;14(22):5663. [CrossRef]
Yang H, Zhang Z, Zhao K, Zhang Y, Yin X, Zhu G, et al. Targeting the adenosine signaling pathway in macrophages for cancer immunotherapy. Hum Immunol. 2024;85(3):110774. [CrossRef]
Geduk A, Atesoglu EB, Mehtap O, Demirsoy ET, Menguc MU, Tarkun P, et al. Programmed cell death ligand 1 expression level and prognostic significance in acute myeloid leukemia. Indian J Hematol Blood Transfus. 2022;38(3):464-72. [CrossRef]
Kurago Z, Guo G, Shi H, Bollag RJ, Groves MW, Byrd JK, et al. Inhibitors of the CD73-adenosinergic checkpoint as promising combinatory agents for conventional and advanced cancer immunotherapy. Front Immunol. 2023;14:1212209. [CrossRef]
Knaus HA, Berglund S, Hackl H, Blackford AL, Zeidner JF, Montiel-Esparza R, et al. Signatures of CD8+ T cell dysfunction in AML patients and their reversibility with response to chemotherapy. JCI Insight. 2018;3(21):e120974. [CrossRef]
Kazerani M, Marcinek A, Philipp N, Brauchle B, Taylor JJ, Moreno HD, et al. Evolution of T-cell fitness through AML progression: enhanced bispecific T-cell engager-mediated function of bone marrow T cells at remission compared to initial diagnosis and relapse. Leukemia. 2024;38(10):2270-5. [CrossRef]
Kong Y, Jia B, Zhao C, Claxton DF, Sharma A, Annageldiyev C, et al. Downregulation of CD73 associates with T cell exhaustion in AML patients. J Hematol Oncol. 2019;12(1):40. [CrossRef]
Brauneck F, Haag F, Woost R, Wildner N, Tolosa E, Rissiek A, et al. Increased frequency of TIGIT+CD73-CD8+ T cells with a TOX+ TCF-1low profile in patients with newly diagnosed and relapsed AML. Oncoimmunology. 2021;10(1):1930391. [CrossRef]
Jiang T, Xu X, Qiao M, Li X, Zhao C, Zhou F, et al. Comprehensive evaluation of NT5E/CD73 expression and its prognostic significance in distinct types of cancers. BMC Cancer. 2018;18(1):267. [CrossRef]
Li Z, Liu H, Ma G, Zhang S, Liu C, Li K, et al. Expression and prognostic impact of CD73 in classical Hodgkin lymphoma [Preprint]. Research Square; 2024 [cited November 3, 2024]. Available from: https://www.researchsquare.com/article/rs-4440165/v1
Ren ZH, Lin CZ, Cao W, Yang R, Lu W, Liu ZQ, et al. CD73 is associated with poor prognosis in HNSCC. Oncotarget. 2016;7(38):61690-702. [CrossRef]
Wettstein MS, Buser L, Hermanns T, Roudnicky F, Eberli D, Baumeister P, et al. CD73 predicts favorable prognosis in patients with nonmuscle-invasive urothelial bladder cancer. Dis Markers. 2015;2015:785461. [CrossRef]
Tripathi A, Lin E, Xie W, Flaifel A, Steinharter JA, Stern Gatof EN, et al. Prognostic significance and immune correlates of CD73 expression in renal cell carcinoma. J Immunother Cancer. 2020;8(2):e001467. [CrossRef]
Zhang JZ, Liu M, Huang ZP. [Expression and clinical significance of CD73 in acute myeloid leukemia patients with NPM1 mutation]. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2021;29(2):416-21. Chinese. [CrossRef]
Gheler FV, Cappellari AR, Renck D, de Souza JB, de Melo RO, Moehlecke BZ, et al. AMP hydrolysis reduction in blood plasma of breast cancer elderly patients after different treatments. Mol Cell Biochem. 2021;476(10):3719-27. [CrossRef]
Wennerberg E, Spada S, Rudqvist NP, Lhuillier C, Gruber S, Chen Q, et al. CD73 blockade promotes dendritic cell infiltration of irradiated tumors and tumor rejection. Cancer Immunol Res. 2020;8(4):465-78. [CrossRef]
Supernat A, Markiewicz A, Welnicka-Jaskiewicz M, Seroczynska B, Skokowski J, Sejda A, et al. CD73 expression as a potential marker of good prognosis in breast carcinoma. Appl Immunohistochem Mol Morphol. 2012;20(2):103-7. [CrossRef]
Messaoudi N, Cousineau I, Arslanian E, Henault D, Stephen D, Vandenbroucke-Menu F, et al. Prognostic value of CD73 expression in resected colorectal cancer liver metastasis. Oncoimmunology. 2020;9(1):1746138. [CrossRef]
Botticelli A, Cirillo A, d'Amati G, Di Gioia C, Corsi A, Della Rocca C, et al. The role of CD73 in predicting the response to immunotherapy in head and neck cancer patients. Pathol Res Pract. 2024;260:155415. [CrossRef]
VOLUME
,
ISSUE
Correspondence
Received
Accepted
Published
Suggested Citation
DOI
License